Medically reviewed by L. Anderson, PharmD Last updated on Dec 11, 2018.
Generic Name:tramadol (TRAM a dol)
Brand Names:ConZip, Ultram
Maybe you've heard that tramadol is a 'safer' pain medication. But is that really true?
The facts: tramadol is a centrally-acting, oral analgesic (pain drug) that contains an opioid (narcotic). Other opioids include drugs you may be more familiar with, like oxycodone or codeine. Opioids have made headlines over the past few years due to the tremendous problem of opioid addiction in the U.S.
Tramadol is approved for the treatment of pain in adults that is severe enough to require an opioid analgesic and for which other treatments do not work or are not tolerated. In addition to acting at the opioid pain receptor, tramadol also inhibits uptake of two neurotransmitters, norepinephrine and serotonin, which may add to its pain-relief effects, although the exact mechanism isn't exactly known.
In 1995, tramadol was originally approved by the U.S. Food and Drug Administration (FDA) as a non-controlled analgesic. However, since 1995, changes to the controlled substance status of tramadol have been made due to reports of drug abuse, misuse and criminal diversion (shifting of any legally prescribed controlled substance from the patient to another person for any illicit use, such as abuse or sale on the streets).
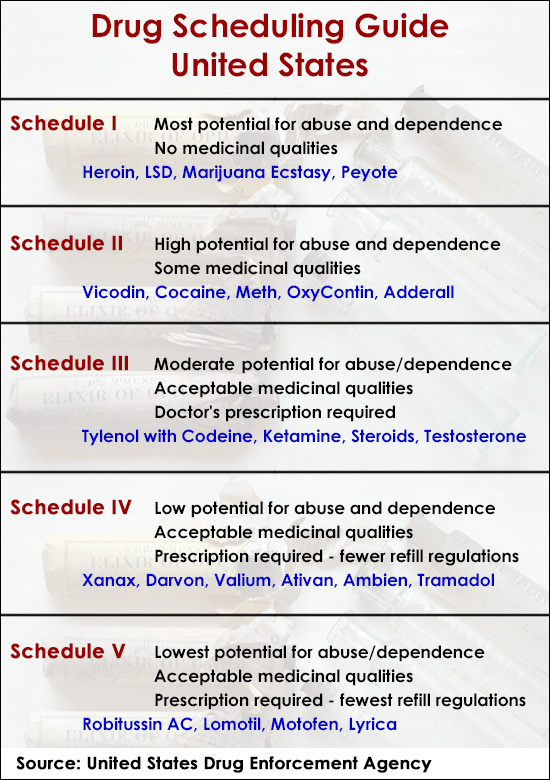
1. Tramadol is now a controlled substance in all 50 U.S. states
The U.S. Drug Enforcement Administration (DEA) announced that tramadol was placed into schedule IV of the Controlled Substances Act (CSA) effective August 18, 2014.
- The new scheduling applies to all forms of tramadol.
- The rescheduling of tramadol came at a time of growing concern related to abuse, misuse, addiction and overdose of opioid analgesics.
- Previously tramadol was a controlled substance in only a few states.
Tramadol prescriptions in the U.S. may now only be refilled up to five times within a six month period after the date on which the prescription was written. After five refills or after six months, whichever occurs first, a new prescription is required. This rule applies to all controlled substances in schedule III and IV.
Ask your pharmacist how to properly dispose of tramadol or any opioid that you are prescribed.
Learn More: How to Safely Dispose of Your Old Medications
2. Tramadol is associated with a wide array of side effects
In many people, tramadol is well-tolerated when used for pain, but tramadol can also cause some common and serious side effects. In fact, the Drug Abuse Warning Network (DAWN) has reported that over 50,000 emergency department visits were related to tramadol use, and over half of these visits were related to side effects of the drug.
It is important to review with your doctor the side effects that may occur with tramadol before starting treatment. Side effects with tramadol may worsen with higher doses or with drug interactions. Starting tramadol slowly and at a lower dose may help to lessen side effects at the beginning of treatment. Call your healthcare provider if you have any of these side effects that are severe or concerning to you.
Common side effects may include:
- headache
- itching
- nausea
- vomiting
- constipation (can be more common in the elderly > 75 years)
- diarrhea
- heartburn
- dizziness
- sleepiness
- nervousness, anxiety, agitation
- tiredness
- stomach pain
Serious side effects, some of which are rare, may include:
- addiction, abuse, and misuse, which may lead to overdose and death, even at normal doses
- depressed breathing (respiratory depression), which may be life-threatening or fatal
- ultra-rapid metabolism (break down of the drug in the body for elimination) of tramadol and other risk factors for life-threatening respiratory depression in children (some cases occurred after tonsillectomy or removal of adenoids).
- neonatal opioid withdrawal syndrome
- effects due to drug interactions with benzodiazepines or other sedative-type (CNS depressant) drugs
- serotonin syndrome
- seizures
- suicide or attempted suicide
- adrenal insufficiency
- severe hypotension (low blood pressure)
- gastrointestinal adverse reactions
- androgen deficiency
- abnormal heart rhythms
- severe hypersensitivity (allergic) reactions
- withdrawal
Breathing changes like slowed or stopped breathing can occur at any time with tramadol, but the risk is greatest when treatment is first started (within the first 24 to 72 hours) or when there is a change in your dose.
You should not use tramadol if have severe asthma or breathing or lung problems, a bowel blockage or narrowing, or an allergy to tramadol. Do not use tramadol if you have taken a monoamine oxidase inhibitor (MAOI), a type of drug for depression, in the last 14 days.
Seizures have occurred in patients taking recommended doses but are more likely at high doses associated with abuse of tramadol.
Do not abruptly stop taking tramadol as withdrawal symptoms like nausea, diarrhea, anxiety, or tremors may occur. Consult with your doctor for a slow, tapering dose schedule if you are stopping tramadol treatment.
Tramadol should not be used during breastfeeding; tell your doctor if you are pregnant or plan to become pregnant.
Warnings: Tramadol Use in Children
- Life-threatening respiratory depression (difficult, slowed breathing) and death have occurred in children who received tramadol. Accidental ingestion of tramadol in children can be fatal.
- Tramadol (brand names include Ultram, ConZip) should NOT be used in children younger than 12 years of age.
- It's especially important that tramadol NOT be used in children up to age 18 years of age after tonsillectomy and/or adenoidectomy surgical procedures (removal of tonsils and/or adenoids).
- Avoid the use of tramadol in adolescents 12 to 18 years of age who have other risk factors that may increase their sensitivity to the respiratory depressant effects of tramadol. Risk factors may include: respiratory depression after surgery, obstructive sleep apnea, obesity, severe lung disease, neuromuscular disease, and use of other medications at the same time as tramadol that also cause respiratory depression (slowed or stopped breathing).
Warnings: Tramadol Use in the Elderly
- The elderly may be especially prone to side effects with tramadol due to lowered liver or kidney function and reduced metabolism or excretion.
- Patients older than 75 years appear to be more prone to gastrointestinal side effects like constipation (30%) than patients less than 65 years (17%).
3. Dangerous metabolism, drug interactions with tramadol
You probably already suspected that tramadol has drug interactions, but you may not know the extent and seriousness that some of these drug interactions can cause. Also, the way that the drug is broken down and excreted from the body (metabolism) is complicated and sometimes unpredictable, especially in children.
If you are known to be an 'ultra-rapid metabolizer' you should not use tramadol. This means that you create the active M1 metabolite from tramadol more quickly than others, and are at risk for dangerous or even fatal respiratory depression or overdose.
Tramadol is known to interact with drugs that are affected by cytochrome P450 (CYP450) enzymes. Specifically it can interact with drugs that affect the 3A4 and 2D6 enzymes. There are hundreds of these drugs, and it's important you ask your pharmacist to check for drug interactions.
- Use of tramadol with these agents can decrease OR increase the metabolism (break down and excretion) of tramadol or M1 in your body, leading to abnormally high or low blood levels of the drug.
- When blood levels are too high, symptoms of opioid toxicity and worsened side effects can occur.
- When blood levels are too low, opioid withdrawal and lack of pain control can occur.
- Drugs like ketoconazole, erythromycin, rifampin, St. John's Wort, or carbamazepine may alter the blood levels of tramadol, but there are many others.
Serious side effects including seizures and serotonin syndrome may also occur due to drug interactions. Examples of drug classes where this might occur include the serotonin reuptake inhibitors (SSRIs, SNRIs), TCAs and MAO inhibitors (like phenelzine or linezolid) -- all types of antidepressants. In fact, tramadol should never be used with an MAOI inhibitor or within 14 days of taking an MAOI. Taking tramadol with drugs that already have a seizure risk may worsen that risk.
Patients receiving serotonergic drugs (for example, the migraine class called “triptans”) may also be at a higher risk for serotonin syndrome.
- Brand names of triptans include: Imitrex, Zomig, Maxalt, and others.
- However, serotonin syndrome and elevated seizure risk can occur with many other medications, too.
- A drug review by your pharmacist can predict if you may be at risk for these dangerous effects.
The use of tramadol with benzodiazepines (or any other sedative, hypnotic or tranquilizer), anti-anxiety medication, muscle relaxant, anesthetic, antipsychotic, alcohol or other narcotic medications (including illegal drugs) can lead to extreme sedation, slowed or stopped breathing, coma and death.
- Tramadol should not be combined with any CNS depressant-type drug unless under the direction of your doctor.
- Do not drink alcohol or use illegal drugs while taking tramadol.
- You should avoid driving, operating machinery or other activities that require mental alertness until the effects of the drug are known and you are sure you can safely drive. If the drug continues to impair your physical or mental ability, do not drive.
Patients should always have a drug interaction review completed each time they start a new medication, or even stop a medication, and that includes prescription drugs, herbals, over-the-counter (OTC) medicines, and supplements like vitamins. Your pharmacist can advise you on possible tramadol drug interactions, so be sure to ask.
4. Tramadol can be habit-forming
Tramadol is structurally related to the opioids like codeine and morphine and can lead to psychological and physical dependence, addiction, and withdrawal. People with a history of a drug-seeking behavior may be at greater risk of addiction, but illicit actions to obtain the drug can occur in people without a prior addiction, as well. Takes steps to secure your tramadol in a safe place at home to prevent theft or accidental overdose.
Do not abruptly stop taking tramadol as withdrawal symptoms like nausea, diarrhea, anxiety, sweating, difficulty in sleep, shivering, pain, tremors, or rarely, hallucinations may occur.
Consult with your doctor before discontinuing tramadol treatment; do NOT discontinue treatment on your own. Withdrawal symptoms may be relieved by re-initiation of opioid therapy followed by a slow, dose reduction combined with symptomatic support, as directed by your doctor.
Learn More: Prescription Drug Addiction: Top Facts for You & Your Family
5. What is an Opioid Analgesic REMS?
Some medications have an inherent risk that require a restricted program known as a Risk Evaluation and Mitigation Strategy (REMS) to ensure safe use. All opioid drugs like tramadol have an Opioid Analgesic REMS program in place as mandated by the FDA.
- A REMS is a medication safety program for certain medications with serious safety concerns or dangerous side effects to help ensure the benefits of the medication outweigh its risks. The FDA may require these and they are developed by the drug manufacturer.
- There are typically many steps to complete for a REMS, and can involve education and certification of healthcare providers, patients, pharmacies, and drug wholesalers.
- Creation of a Medication Guide that is updated with important patient information is part of the Opioid Analgesic REMS. The patient receives this each time they fill their opioid prescription and should review for changes.
6. Tramadol is available in both immediate-release and extended-release formulations.
Both the immediate-release and extended-release formulation of tramadol are available generically and can possibly save you hundreds of dollars on your prescription.
If you prefer generic medications due to cost-savings, ask your physician to only write for generic drugs whenever possible. If you cannot afford your medication, do not walk away from the pharmacy. Ask your doctor or pharmacist for more affordable alternatives.
The extended-release form of tramadol is for around-the-clock treatment of pain and not for use on an as-needed basis. Tramadol extended-release tablets must be taken whole, and not split, chewed or crushed.
A combination product of tramadol and acetaminophen (Ultracet) is also available by prescription in both brand and generic form.
- Combining tramadol with acetaminophen (Tylenol) may further help with pain and allow lower doses of tramadol to be used.
- However, Ultracet is not approved for use for more than 5 days.
- Also, be careful not to combine Ultracet with extra acetaminophen (Tylenol), as too much acetaminophen is toxic for your liver.
Learn more about generic drugs in “Facts About Generic Drugs”.
7. Dose adjustments are needed in the elderly, and in those with kidney or liver problems.
Here's another good reason not to share your tramadol with others: the dose you are prescribed may not be the right dose for someone else, and could lead to serious consequences like slowed or stopped breathing, coma, or even death.
As with many medications, if you are young, elderly, or have kidney or liver disease dose adjustments are often required.
- The dosing interval (how often you take the drug) may need to be adjusted, the actual dose of the drug may need to be reduced, and you may have a maximum daily dose you should not exceed.
- Talk to your doctor about the need for adjusted doses with any medication, including tramadol.
- Do not exceed the prescribed dose or dosing interval prescribed by your doctor.
Patients older than 65 years of age
- Doses should usually start at the low end of the dosing range and can be titrated upwards slowly based on tolerance and effectiveness.
Patients older than 75 years of age
- Maximum dose of regular-release tramadol oral tablets: 300 mg per day (in divided doses).
Kidney Disease and Liver Disease
Over 30% of tramadol is excreted by the kidneys as the unchanged molecule, which means it could lead to toxic blood levels in patients with kidney disease.
- Tramadol is metabolized through the liver, so a patient with kidney or liver disease may require a change in dose, different dose interval, or maximum amount of drug taken each day.
- Some patients with severe kidney or liver impairment may not be candidates for tramadol treatment at all.
- Providers should consider monitoring renal function (such as creatinine clearance) with tramadol treatment, especially in the elderly, because this drug is substantially excreted by the kidney.
8. There are ways to engage with other patients using tramadol.
Support groups may be helpful for patients who take tramadol, who use medications for pain relief, who are in need of addiction support, and for many other needs.
Joining one or more support groups is a great way to discover others taking related medications with similar medical conditions, keep up with the news, and share your own experience.
- Ultram Support Group
There are over 1300 reviews for tramadol from patients who use this drug for general pain, back pain, tooth pain and other various conditions (some of which may be off-label use, meaning the drug is not approved by the FDA for that particular use).
Remember, this information is NOT intended to endorse tramadol or recommend therapy. While these reviews may or may not be helpful to you, they are NOT a substitute for the expertise, skill, knowledge and judgement of your individual healthcare provider.
Sources
- Ultram product label. Janssen Pharmaceuticals, Inc. Revised 9/2018. Accessed Dec. 11, 2018 at https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/020281s042s043lbl.pdf
- Drug Enforcement Administration. Office of Diversion Control. Drug & Chemical Evaluation Section. Tramadol. (Trade Names: Ultram, Ultracet).
- Bush DM. Emergency Department Visits for Adverse Reactions Involving the Pain Medication Tramadol. 2015 May 14. In: The CBHSQ Report. Rockville (MD): Substance Abuse and Mental Health Services Administration (US); 2013. Accessed Dec. 11, 2018 at https://misuse.ncbi.nlm.nih.gov/error/abuse.shtml
- Drug Enforcement Administration. Office of Diversion Control. Drug and Chemical Evaluation Section. Tramadol.
(Trade Names: Ultram, Ultracet). Accessed Feb. 27, 2018. - Sansone RA, Sansone LA. Tramadol: Seizures, Serotonin Syndrome, and Coadministered Antidepressants. Psychiatry (Edgmont). 2009;6:17-21. Accessed Feb. 27, 2018 at https://misuse.ncbi.nlm.nih.gov/error/abuse.shtml
- Tramadol Product Label. Drugs.com. Accessed Dec. 12, 2018 at https://www.drugs.com/tramadol.html
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
§ 25.72. Schedules of controlled substances.
(a) General. In accordance with sections 3 and 4 of the act (35 P. S.
§ § 780-103 and 780-104), this section lists all controlled substances. Section 4 of the act (35 P. S. § 780-104) designates specific substances for inclusion under the five schedules. The substances listed in this section include those listed by section 4 of the act (35 P. S. § 780-104) and those that have been added by the Secretary after consultation with the Drug, Device and Cosmetic Board.
(b) Schedule I. In determining that a substance comes within this schedule, the Secretary will find: a high potential for abuse; no currently accepted medical use in the United States; and a lack of accepted safety for use under medical supervision. The following controlled substances are included in this schedule:
(1) The following opiates, including their isomers, esters, ethers, salts, and salts of isomers, esters, and ethers, unless specifically excepted whenever the existence of the isomers, esters, ethers, and salts is possible within the specific chemical designation:
(i) [Reserved].
(ii) Allylprodine.
(iii) Alphacetylmethadol.
(iv) Alphameprodine.
(v) Alphamethadol.
(v.1) Alpha-methylfentanyl.
(vi) Benzethidine.
(vii) Betacetylmethadol.
(viii) Betameprodine.
(ix) Betamethadol.
(x) Betaprodine.
(xi) Clonitazene.
(xii) Dextromoramide.
(xiii) Dextrorphan (except its methylether).
(xiv) Diampromide.
(xv) Diethylthiambutene.
(xvi) Dimenoxadol.
(xvii) Dimepheptanol.
(xviii) Dimethylthiambutene.
(xix) Dioxaphetyl butyrate.
(xx) Dipipanone.
(xxi) Ethylmethylthiambutene.
(xxii) Etonitazene.
(xxiii) Etoxeridine.
(xxiv) Furethidine.
(xxv) Hydroxypethidine.
(xxvi) Ketobemidone.
(xxvii) Levomoramide.
(xxviii) Levophenacylmorphan.
(xxix) Morpheridine.
(xxx) Noracymethadol.
(xxxi) Norlevorphanol.
(xxxii) Normethadone.
(xxxiii) Norpipanone.
(xxxiv) Phenadoxone.
(xxxv) Phenampromide.
(xxxvi) Phenomorphan.
(xxxvii) Phenoperidine.
(xxxviii) Piritramide.
(xxxix) Proheptazine.
(xl) Properidine.
(xli) Racemoramide.
(xlii) Tilidine.
(xliii) Trimeperidine.
(xliv) [Reserved].
(xlv) 3-Methylfentanyl.
(xlvi) 1-Methyl-4-Phenyl-4-Propionoxypiperidine (MPPP).
(xlvii) 1-(2-Phenylethyl)-4-Phenyl-4-etyloxypiperidine (PEPAP).
(xlviii) Para-fluorofentanyl.
(xlix) Acetyl-alpha-methylfentanyl.
(l) Alph-methylthiofentanyl.
(li) Beta-hydroxyfentanyl.
(lii) 3 Methylthiofentanyl.
(liii) Thiofentanyl.
(liv) Beta-hydroxy-3-methylfentanyl.
(2) The following opium derivatives, their salts, isomers and salts of isomers, unless specifically excepted, whenever the existence of the salts, isomers, and salts of isomers is possible within the specific chemical designation:
(i) Acetorphine.
(ii) Acetyldihydrocodeine.
(iii) Benzylmorphine.
(iv) Codeine methylbromide.
(v) Codeine-N-Oxide.
(vi) Cyprenorphine.
(vii) Desomorphine.
(viii) Dihydromorphine.
(ix) Drotebanol (added August 6, 1978).
(x) Etorphine.
(xi) Heroin.
(xii) Hydromorphinol.
(xiii) Methyldesorphine.
(xiv) Methylhydromorphine.
(xv) Morphine methylbromide.
(xvi) Morphine methylsulfonate.
(xvii) Morphine-N-Oxide.
(xviii) Myrophine.
(xix) Nicocodeine.
(xx) Nicomorphine.
(xxi) Normorphine.
(xxii) Pholcodine.
(xxiii) Thebacon.
(3) A material, compound, mixture or preparation which contains any quantity of the following hallucinogenic substances, their salts, isomers, and salts of isomers, unless specifically excepted, whenever the existence of the salts, isomers, and salts of isomers is possible within the specific chemical designation:
(i) 3,4-methylenedioxy amphetamine.
(ii) 5-methoxy-3, 4-methylenedioxy amphetamine.
(iii) 3,4,5-trimethoxy amphetamine.
(iv) Bufotenine.
(v) Diethyltryptamine.
(vi) Dimethyltryptamine.
(vii) 4-methyl-2, 5-dimethoxyamphetamine.
(viii) Ibogaine.
(ix) Lysergic acid diethylamide.
(x) Mescaline.
(xi) Peyote.
(xii) N-ethyl-3-piperidyl benzilate.
(xiii) N-methyl-3-piperidyl benzilate.
(xiv) Psilocybin.
(xv) Psilocyn.
(xvi) Tetrahydrocannabinols.
(xvii) 3, 4-methylenedioxy-N-ethylamphetamine.
(xviii) N-hydroxy-3, 4-methylenedioxyamphetamine.
(xix) 2, 5-Dimethoxy-4-ethylamphetamine (DOET).
(xx) 4 Bromo 2, 5 Dimethoxyphenethylamine.
(4) Marihuana.
(5) 4-Bromo-2, 5 Dimethoxyamphetamine (4-Bromo, 2, 5 DMA) (added October 17, 1975).
(6) Unless specifically excepted or unless listed in another schedule, a material, compound, mixture or preparation which contains any quantity of the following substances including the salts, isomers and salts of isomers:
(i) Fenethylline.
(ii) N-ethylamphetamine.
(iii) Methaqualone.
(iv) Bromazepam.
(v) Camazepam.
(vi) Clotiazepam.
(vii) Cloxazolam.
(viii) Delorazepam.
(ix) Ethyl loflazepate.
(x) Fludiazepam.
(xi) Flunitrazepam.
(xii) Haloxazolam.
(xiii) Ketazolam.
(xiv) Loprazolam.
(xv) Lormetazepam.
(xvi) Medazepam.
(xvii) Nimetazepam.
(xiii) Nitrazepam.
(xix) Nordiazepam.
(xx) Oxazolam.
(xxi) Pinazepam.
(xxii) Tetrazepam.
(xxiiii) 3, 4-Methylenedioxymethamphetamine (MDMA)
(xxiv) 4-methylaminorex.
(xxv) Cathinone.
(xxvi) Methcathinone HCL.
(xxvii) Dimethylamphetamine.
(xxiii) 1-(3-trifluoromethylphenyl) Piperazine (TFMPP)
(xxix) N-Benzylpiperazine (BZP)
(xxx) Alpha-Methyltryptamine (AMT)
(xxxi) 2-5 Dimethoxy-4-(N)-Propylthiophenethylamine (2C-T-7)
(xxxii) 5-Methoxy-N, N-Diisopropyltryptamine (5-MEO-DIPT)
(c) Schedule II. In determining that a substance comes within this schedule, the Secretary will find: a high potential for abuse; currently accepted medical use in the United States; or currently accepted medical use with severe restrictions and abuse may lead to severe psychic or physical dependence. The following controlled substances are included in this schedule:
(1) The following substances of any quantity, except those narcotics specifically excepted or listed in other schedules, whether produced directly or indirectly by extraction from substances of vegetable origin, or independently by means of chemical synthesis, or by combination of extraction and chemical synthesis:
(i) Opium and opiate, and a salt, compound, derivative or preparation of opium or opiate.
(ii) A salt, compound, derivative or preparation thereof which is chemically equivalent or identical with the substances referred to in subparagraph (i) except that these substances may not include the isoquinoline alkaloids of opium.
(iii) Opium poppy and poppy straw.
(iv) Coca leaves and a salt, compound, derivative or preparation of coca leaves, and a salt, compound, derivative or preparation thereof which is chemically equivalent or identical with these substances, but may not include decocanized coca leaves or extracts of coca leaves, which extracts do not contain cocaine or ecgonine.
(2) The following opiates, including their isomers, esters, ethers, salts and salts of isomers, esters and ethers, of any quantity, unless specifically excepted or listed in another schedule, whenever the existence of the isomers, esters, ethers and salts is possible within the specific chemical designation:
(i) Alphaprodine.
(ii) Anileridine.
(iii) Bezitramide.
(iv) Dihydrocodeine.
(v) Diphenoxylate.
(vi) Fentanyl.
(vii) Isomethadone.
(viii) Levomethorphan.
(ix) Levorphanol.
(x) Metazocine.
(xi) Methadone.
(xii) Methadone-Intermediate, 4-cyano-2-dimethylamino-4, 4-diphenyl butane.
(xiii) Moramide-Intermediate, 2-methyl-3-morpholino-1, 1-diphenylpropane-carboxylicacid.
(xiv) Pethidine.
(xv) Pethidine-Intermediate-A, 4-cyano-1-methyl-4-phenylpiperidine.
(xvi) Pethidine-Intermediate-B, ethyl-4-phenylpiperidine-4-carboxylate.
(xvii) Pethidine-Intermediate-C, 1-methyl-4-phenylpiperidine-4-carboxylic acid.
(xviii) Phenazocine.
(xix) Piminodine.
(xx) Propiram (added August 5, 1978).
(xxi) Racemethorphan.
(xxii) Racemorphan.
(xxiii) Sufentanil.
(xxiv) Alfentanil.
(xxv) Carfentanil.
(xxvi) Levo-Alpha Acetyl-Methadol.
(3) Unless specifically excepted or unless listed in another schedule, a material, compound, mixture or preparation which contains any quantity of the following substances:
(i) Amphetamine, its salts, optical isomers and salts of its optical isomers.
(ii) Phenmetrazine and its salts.
(iii) Methylphenidate.
(iv) Methamphetamine including its salts, isomers and salts of isomers.
(v) Phenylacetone.
(vi) Nabilone.
(vii) Glutethimide.
(4) The phrase ‘‘opiates’’ as used in section 4 of the act (35 P. S.
§ 780-104) and elsewhere throughout the act may not include the dextrorotatory isomer of 3-methoxy-n-methylmorphinan and its salts, but does include its racemic and levorotatory forms.
(5) A material, compound, mixture or preparation, unless specifically excepted, which contains a quantity of:
(i) Phencyclidine.
(ii) 1-phenylcyclohexylamine.
(iii) 1-piperidinocyclohexanecarbonitrile.
(iv) Nabilone.
(6) Unless specifically excepted or unless listed in another schedule, any material, compound, mixture or preparation which contains any quantity of the following substances, including its salts, isomers and salts of isomers whenever the existence of the salts, isomers and salts of isomers is possible within the specific chemical designation:
(i) Amobarbital (added August 21, 1976).
(ii) Secobarbital (added August 21, 1976).
(iii) Pentobarbital (added August 21, 1976).
(d) Schedule III. In determining that a substance comes within this schedule, the Secretary will find: a potential for abuse less than the substances listed in Schedules I and II; well documented and currently accepted medical use in the United States; and abuse may lead to moderate or low physical dependence. The following classes of controlled substances are included in this schedule:
(1) A material, compound, mixture or preparation unless specifically excepted or unless listed in another schedule which contains any quantity of the following substances:
(i) A substance which contains any quantity of a derivative of barbituric acid, or a salt of a derivative of barbituric acid.
(ii) Chorhexadol.
(iii) Lysergic acid.
(iv) Lysergic acid amide.
(v) Methyprylon.
(vi) Sulfondiethylmethane.
(vii) Sulfonethylmethane.
(viii) Sulfonmethane.
(2) Nalorphine.
(3) A material, compound, mixture, or preparation containing limited quantities of the following narcotic drugs, or salts thereof, unless specifically excepted or listed in other schedules.
(i) Not more than 1.8 grams of codeine per 100 milliliters or not more than 90 milligrams per dosage unit, with an equal or greater quantity of an isoquinoline alkaloid of opium.
(ii) Not more than 1.8 grams of codeine per 100 milliliters or not more than 90 milligrams per dosage unit, with one or more active, nonnarcotic ingredients in recognized therapeutic amounts.
(iii) Not more than 300 milligrams of dihydrocodeinone per 100 milliliters or not more than 15 milligrams per dosage unit, with a fourfold or greater quantity of an isoquinolene alkaloid of opium.
(iv) Not more than 300 milligrams of dihydrocodeinone per 100 milliliters or not more than 15 milligrams per dosage unit, with one or more active, nonnarcotic ingredients in recognized therapeutic amounts.
(v) Not more than 1.8 grams of dihydrocodeine per 100 milliliters or not more than 90 milligrams per dosage unit, with one or more active, nonnarcotic ingredients in recognized therapeutic amounts.
(vi) Not more than 300 milligrams of ethylmorphine per 100 milliliters or not more than 15 milligrams per dosage unit, with one or more active, nonnarcotic ingredients in recognized therapeutic amounts.
(vii) Not more than 500 milligrams of opium per 100 milliliters or per 100 grams, or not more than 2.5 milligrams per dosage unit with one or more active, nonnarcotic ingredients in recognized therapeutic amounts.
(4) Unless specifically excepted or unless listed in another schedule, a material, compound, mixture, or preparation which contains any quantity of the following substances including its salts, isomers, whether optical position or geometric, and salts of the isomers whenever the existence of the salts, isomers, and salts of isomers is possible within the specific chemical designation, Schedule III shall include the following:
(i) Benzphetamine (added August 21, 1976).
(ii) Chlorphentermine (added August 21, 1976).
(iii) Clortermine (added August 21, 1976).
(iv) [Reserved].
(v) Phendimetrazine (added August 21, 1976).
(5) A compound, mixture or preparation or a salt thereof including one or more other active medicinal ingredients which are not listed in a schedule containing the following:
(i) Amobarbital.
(ii) Secobarbital.
(iii) Pentobarbital.
(6) A suppository dosage form or a salt thereof approved by the Food and Drug Administration for marketing only as a suppository containing the following:
(i) Amobarbital.
(ii) Secobarbital.
(iii) Pentobarbital.
(7) The Secretary may, by regulation, except a compound, mixture, or preparation containing a drug or controlled substance listed in this schedule from the application of those provisions of the act covering controlled substances, if the compound, mixture, or preparation contains one or more active medicinal ingredients not having a stimulant or depressant effect on the central nervous system; provided, that the admixtures shall be included therein in the combinations, quantity, proportion, or concentration as to vitiate the potential for abuse of the substances which do have a stimulant or depressant effect on the central nervous system.
(8) The Secretary will, by regulation, exempt a nonnarcotic substance from the control under the act if the substance may, under the provisions of the Federal Food, Drug, and Cosmetic Act (21 U.S.C.A. § 301 et seq.), be lawfully sold over the counter without a prescription.
(9) A material, compound, mixture or preparation, unless specifically excepted, which contains a quantity of Dronabinol—synthetic—in sesame oil encapsulated in a soft gelatin capsule but only those drug products approved by the United States Food and Drug Administration.
(10) Buprenorphine.
(e) Schedule IV. In determining that a substance comes within this schedule, the Secretary will find: a low potential for abuse relative to substances in Schedule III; currently accepted medical use in the United States; and limited physical or psychological dependence liability relative to the substances listed in Schedule III. The following controlled substances are included in this schedule:
(1) A material, compound, mixture or preparation, unless specifically excepted or unless listed in another schedule, which contains a quantity of the following substances:
(i) Barbital.
(ii) Chloral betaine.
(iii) Chloral hydrate.
(iv) Ethchlorvynol.
(v) Ethinamate.
(vi) Methohexital.
(vii) Meprobamate.
(viii) Methylphenobarbital.
(ix) Paraldehyde.
(x) Petrichloral.
(xi) Phenobarbital.
(xii) Chlordiazepoxide (added August 21, 1976).
(xiii) Diazepam (added August 21, 1976).
(xiv) Oxazepam (added August 21, 1976).
(xv) Clorazepate (added August 21, 1976).
(xvi) Flurazepam (added August 21, 1976).
(xvii) Clonazepam (added August 21, 1976).
(xviii) Mebutamate (added August 21, 1976).
(xix) Temazepam.
(xx) Alprazolam.
(xxi) Halazepam.
(xxii) Triazolam.
(xxiii) Midazolam.
(xxiv) Quazepam.
(xxv) Estazolam.
(xxvi) Zolpidem.
(xxvii) Clobazam. (added March 27, 2012)
(2) A material, compound, mixture, or preparation which contains any quantity of the following substance including its salts, isomers whether optical position or geometric, and salts of the isomers, whenever the existence of the salts, isomers, and salts of isomers is possible:
(i) Fenfluramine (added August 21, 1976).
(ii) Pentazocine (added January 19, 1980).
(iii) Lorazepam (added January 19, 1980).
(iv) Prazepam (added January 19, 1980).
(v) Dextropropoxyphene (added January 19, 1980).
(3) Unless specifically excepted or unless listed in another schedule, a material, compound, mixture or preparation which contains any quantity of the following substances including its salts, isomers whether optical position or geometric, and salts of the isomers whenever the existence of the salts, isomers and salts of isomers is possible within the specific chemical designation:
(i) Diethylpropion (added August 21, 1976).
(ii) Phentermine (added August 21, 1976).
(iii) Pemoline (added August 21, 1976).
(iv) Mazindol.
(v) Pipradol.
(vi) SPA (1-dimethylamino-1-2-diphenylethane).
(vii) Cathine.
(viii) Fencamfamin.
Is Tramadol A Schedule 2 Drug Test
(ix) Fenproporex.
(x) Mefenorex.
(xi) Butorphanol.
(xii) Sibutramine.
(4) The Secretary may, by regulation, except a compound, mixture, or preparation containing a drug or controlled dangerous substance listed in paragraph (1) from the application of those provisions of the act, sections 3 and 4 of the act (35 P. S. § § 780-103 and 780-104), covering controlled drugs, if the compound, mixture, or preparation contains one or more active medicinal ingredients not having a stimulant or depressant effect on the central nervous system; provided that the admixtures shall be included therein in combinations, quantity, proportion, or concentration as to vitiate the potential for abuse of the substances which do have a stimulant or depressant effect on the central nervous system.
(5) The Secretary shall by regulation exempt a nonnarcotic substance from the control under the act if the substance may, under the provisions of the Federal Food, Drug, and Cosmetic Act (21 U.S.C.A. § 301 et seq.) be lawfully sold over the counter without a prescription.
(6) A compound, mixture, or preparation which purports to have a cough suppressant effect and which contains a limited quantity of the following narcotics or their salts, and which contains in addition one or more nonnarcotic active medicinal ingredients in sufficient proportion to confer upon the compound, mixture, or preparation, valuable medicinal qualities other than those possessed by the narcotic alone shall be included under this schedule:
(i) Not more than 200 milligrams of codeine, or any of its salts, per 100 milliliters or per 100 grams and not more than 10 milligrams per dosage unit.
(ii) Not more than 100 milligrams of dihydrocodeine, or any of its salts, per 100 milliliters or per 100 grams and not more than 5 milligrams per dosage unit.
(iii) Not more than 100 milligrams of ethylmorphine, or any of its salts, per 100 milliliters or per 100 grams and not more than 5 milligrams per dosage unit.
(f) Schedule V. In determining that a substance comes within this schedule, the Secretary will find: a low potential for abuse relative to the substances listed in Schedule IV; currently accepted medical use in the United States; and limited physical dependence or psychological dependence liability relative to the substances listed in Schedule IV. The following controlled substances are included in this schedule:
(1) A compound, mixture, or preparation containing limited quantities of any of the following narcotics or any of their salts, which shall include one or more nonnarcotic active medicinal ingredients in sufficient proportion to confer upon the compound, mixture, or preparation, valuable medicinal qualities other than those possessed by the narcotic alone:
(i) Not more than 200 milligrams of codeine, or any of its salts, per 100 milliliters or per 100 grams and not more than 10 milligrams per dosage unit.
(ii) Not more than 100 milligrams of dihydrocodeine or any of its salts, per 100 milliliters or per 100 grams and not more than 5 milligrams per dosage unit.
(iii) Not more than 100 milligrams of ethylmorphine, or any of its salts, per 100 milliliters or per 100 grams and not more than 5 milligrams per dosage unit.
(iv) Not more than 2.5 milligrams of diphenoxylate and not less than 25 micrograms of altropine sulfate per dosage unit.
(v) Not more than 100 milligrams of opium per 100 milliliters or per 100 grams, or not more than 5 milligrams per dosage unit.
(2) Propylhexadrine, except when labeled for over-the-counter drug sale in conformity with 21 CFR 1308.15 (relating to schedule V).
(3) Pyrovalerone.
The provisions of this § 25.72 amended under section 2102(g) of The Administrative Code of 1929 (71 P. S. § 532(g)); sections 3(a) and (c) and 4 of The Controlled Substance, Drug, Device and Cosmetic Act (35 P. S. § § 780-103(a) and (c) and 780-104); and section 2 of the Optometric Practice and Licensure Act (63 P. S. § 244.2).
The provisions of this § 25.72 amended February 23, 1979, effective February 24, 1979, 9 Pa.B. 611; amended January 18, 1980, effective January 19, 1980, 10 Pa.B. 216; amended August 13, 1982, effective August 14, 1982, 12 Pa.B. 2685; amended November 4, 1983, effective November 5, 1983, 13 Pa.B. 3376; amended September 7, 1984, effective September 8, 1984, 14 Pa.B. 3250; amended October 26, 1984, effective November 5, 1983, 14 Pa.B. 3909; amended March 15, 1986, effective March 16, 1986, 16 Pa.B. 812; amended August 29, 1986, effective August 30, 1986, 16 Pa.B. 3209; corrected February 13, 1987, 17 Pa.B. 717; amended April 17, 1987, effective April 18, 1987, 17 Pa.B. 1567; amended May 20, 1988, effective May 21, 1988, 18 Pa.B. 2298; amended May 20, 1988, effective May 21, 1988, 18 Pa.B. 2299; corrected July 1, 1988, effective April 16, 1977, 18 Pa.B. 2905; amended February 17, 1989, effective February 18, 1989, 19 Pa.B. 638; amended March 10, 1989, effective March 11, 1989, 19 Pa.B. 997; amended November 21, 1997, effective November 22, 1997, 27 Pa.B. 6088; amended May 4, 2001, effective May 5, 2001, 31 Pa.B. 2373; amended May 21, 2004, effective May 22, 2004, 34 Pa.B. 2711; amended March 17, 2006, effective March 18, 2006, 36 Pa.B. 1218; amended July 10, 2015, effective July 11, 2015, 45 Pa.B. 3708. Immediately preceding text appears at serial pages (236115) to (236116), (317777) to (317778), (279223) to (279224), (304185) to (304188) and (366337).
A material containing the drug cathinone is a controlled substance under § 25.72(b)(6)(xxvi). The language of the § 25.72(b)(6)(xxvi) plainly states that an unlisted material, when containing a controlled substance, is a controlled substance. Commonwealth v. Mohamud, 15 A.3d 80, 92 (Pa. Super. 2010).
This section cited in 28 Pa. Code § 551.3 (relating to definitions); and 49 Pa. Code § 21.284a (relating to prescribing and dispensing drugs).
No part of the information on this site may be reproduced for profit or sold for profit.
This material has been drawn directly from the official Pennsylvania Code full text database. Due to the limitations of HTML or differences in display capabilities of different browsers, this version may differ slightly from the official printed version.
Medically reviewed by L. Anderson, PharmD Last updated on Dec 11, 2018.
Generic Name:tramadol (TRAM a dol)
Brand Names:ConZip, Ultram
Maybe you've heard that tramadol is a 'safer' pain medication. But is that really true?
The facts: tramadol is a centrally-acting, oral analgesic (pain drug) that contains an opioid (narcotic). Other opioids include drugs you may be more familiar with, like oxycodone or codeine. Opioids have made headlines over the past few years due to the tremendous problem of opioid addiction in the U.S.
Tramadol is approved for the treatment of pain in adults that is severe enough to require an opioid analgesic and for which other treatments do not work or are not tolerated. In addition to acting at the opioid pain receptor, tramadol also inhibits uptake of two neurotransmitters, norepinephrine and serotonin, which may add to its pain-relief effects, although the exact mechanism isn't exactly known.
In 1995, tramadol was originally approved by the U.S. Food and Drug Administration (FDA) as a non-controlled analgesic. However, since 1995, changes to the controlled substance status of tramadol have been made due to reports of drug abuse, misuse and criminal diversion (shifting of any legally prescribed controlled substance from the patient to another person for any illicit use, such as abuse or sale on the streets).
1. Tramadol is now a controlled substance in all 50 U.S. states
The U.S. Drug Enforcement Administration (DEA) announced that tramadol was placed into schedule IV of the Controlled Substances Act (CSA) effective August 18, 2014.
- The new scheduling applies to all forms of tramadol.
- The rescheduling of tramadol came at a time of growing concern related to abuse, misuse, addiction and overdose of opioid analgesics.
- Previously tramadol was a controlled substance in only a few states.
Tramadol prescriptions in the U.S. may now only be refilled up to five times within a six month period after the date on which the prescription was written. After five refills or after six months, whichever occurs first, a new prescription is required. This rule applies to all controlled substances in schedule III and IV.
Ask your pharmacist how to properly dispose of tramadol or any opioid that you are prescribed.
Learn More: How to Safely Dispose of Your Old Medications
2. Tramadol is associated with a wide array of side effects
In many people, tramadol is well-tolerated when used for pain, but tramadol can also cause some common and serious side effects. In fact, the Drug Abuse Warning Network (DAWN) has reported that over 50,000 emergency department visits were related to tramadol use, and over half of these visits were related to side effects of the drug.
It is important to review with your doctor the side effects that may occur with tramadol before starting treatment. Side effects with tramadol may worsen with higher doses or with drug interactions. Starting tramadol slowly and at a lower dose may help to lessen side effects at the beginning of treatment. Call your healthcare provider if you have any of these side effects that are severe or concerning to you.
Common side effects may include:
- headache
- itching
- nausea
- vomiting
- constipation (can be more common in the elderly > 75 years)
- diarrhea
- heartburn
- dizziness
- sleepiness
- nervousness, anxiety, agitation
- tiredness
- stomach pain
Serious side effects, some of which are rare, may include:
- addiction, abuse, and misuse, which may lead to overdose and death, even at normal doses
- depressed breathing (respiratory depression), which may be life-threatening or fatal
- ultra-rapid metabolism (break down of the drug in the body for elimination) of tramadol and other risk factors for life-threatening respiratory depression in children (some cases occurred after tonsillectomy or removal of adenoids).
- neonatal opioid withdrawal syndrome
- effects due to drug interactions with benzodiazepines or other sedative-type (CNS depressant) drugs
- serotonin syndrome
- seizures
- suicide or attempted suicide
- adrenal insufficiency
- severe hypotension (low blood pressure)
- gastrointestinal adverse reactions
- androgen deficiency
- abnormal heart rhythms
- severe hypersensitivity (allergic) reactions
- withdrawal
Breathing changes like slowed or stopped breathing can occur at any time with tramadol, but the risk is greatest when treatment is first started (within the first 24 to 72 hours) or when there is a change in your dose.
You should not use tramadol if have severe asthma or breathing or lung problems, a bowel blockage or narrowing, or an allergy to tramadol. Do not use tramadol if you have taken a monoamine oxidase inhibitor (MAOI), a type of drug for depression, in the last 14 days.
Seizures have occurred in patients taking recommended doses but are more likely at high doses associated with abuse of tramadol.
Do not abruptly stop taking tramadol as withdrawal symptoms like nausea, diarrhea, anxiety, or tremors may occur. Consult with your doctor for a slow, tapering dose schedule if you are stopping tramadol treatment.
Tramadol should not be used during breastfeeding; tell your doctor if you are pregnant or plan to become pregnant.
Warnings: Tramadol Use in Children
- Life-threatening respiratory depression (difficult, slowed breathing) and death have occurred in children who received tramadol. Accidental ingestion of tramadol in children can be fatal.
- Tramadol (brand names include Ultram, ConZip) should NOT be used in children younger than 12 years of age.
- It's especially important that tramadol NOT be used in children up to age 18 years of age after tonsillectomy and/or adenoidectomy surgical procedures (removal of tonsils and/or adenoids).
- Avoid the use of tramadol in adolescents 12 to 18 years of age who have other risk factors that may increase their sensitivity to the respiratory depressant effects of tramadol. Risk factors may include: respiratory depression after surgery, obstructive sleep apnea, obesity, severe lung disease, neuromuscular disease, and use of other medications at the same time as tramadol that also cause respiratory depression (slowed or stopped breathing).
Warnings: Tramadol Use in the Elderly
- The elderly may be especially prone to side effects with tramadol due to lowered liver or kidney function and reduced metabolism or excretion.
- Patients older than 75 years appear to be more prone to gastrointestinal side effects like constipation (30%) than patients less than 65 years (17%).
3. Dangerous metabolism, drug interactions with tramadol
You probably already suspected that tramadol has drug interactions, but you may not know the extent and seriousness that some of these drug interactions can cause. Also, the way that the drug is broken down and excreted from the body (metabolism) is complicated and sometimes unpredictable, especially in children.
If you are known to be an 'ultra-rapid metabolizer' you should not use tramadol. This means that you create the active M1 metabolite from tramadol more quickly than others, and are at risk for dangerous or even fatal respiratory depression or overdose.
Tramadol is known to interact with drugs that are affected by cytochrome P450 (CYP450) enzymes. Specifically it can interact with drugs that affect the 3A4 and 2D6 enzymes. There are hundreds of these drugs, and it's important you ask your pharmacist to check for drug interactions.
- Use of tramadol with these agents can decrease OR increase the metabolism (break down and excretion) of tramadol or M1 in your body, leading to abnormally high or low blood levels of the drug.
- When blood levels are too high, symptoms of opioid toxicity and worsened side effects can occur.
- When blood levels are too low, opioid withdrawal and lack of pain control can occur.
- Drugs like ketoconazole, erythromycin, rifampin, St. John's Wort, or carbamazepine may alter the blood levels of tramadol, but there are many others.
Serious side effects including seizures and serotonin syndrome may also occur due to drug interactions. Examples of drug classes where this might occur include the serotonin reuptake inhibitors (SSRIs, SNRIs), TCAs and MAO inhibitors (like phenelzine or linezolid) -- all types of antidepressants. In fact, tramadol should never be used with an MAOI inhibitor or within 14 days of taking an MAOI. Taking tramadol with drugs that already have a seizure risk may worsen that risk.
Patients receiving serotonergic drugs (for example, the migraine class called “triptans”) may also be at a higher risk for serotonin syndrome.
- Brand names of triptans include: Imitrex, Zomig, Maxalt, and others.
- However, serotonin syndrome and elevated seizure risk can occur with many other medications, too.
- A drug review by your pharmacist can predict if you may be at risk for these dangerous effects.
The use of tramadol with benzodiazepines (or any other sedative, hypnotic or tranquilizer), anti-anxiety medication, muscle relaxant, anesthetic, antipsychotic, alcohol or other narcotic medications (including illegal drugs) can lead to extreme sedation, slowed or stopped breathing, coma and death.
- Tramadol should not be combined with any CNS depressant-type drug unless under the direction of your doctor.
- Do not drink alcohol or use illegal drugs while taking tramadol.
- You should avoid driving, operating machinery or other activities that require mental alertness until the effects of the drug are known and you are sure you can safely drive. If the drug continues to impair your physical or mental ability, do not drive.
Patients should always have a drug interaction review completed each time they start a new medication, or even stop a medication, and that includes prescription drugs, herbals, over-the-counter (OTC) medicines, and supplements like vitamins. Your pharmacist can advise you on possible tramadol drug interactions, so be sure to ask.
4. Tramadol can be habit-forming
Tramadol is structurally related to the opioids like codeine and morphine and can lead to psychological and physical dependence, addiction, and withdrawal. People with a history of a drug-seeking behavior may be at greater risk of addiction, but illicit actions to obtain the drug can occur in people without a prior addiction, as well. Takes steps to secure your tramadol in a safe place at home to prevent theft or accidental overdose.
Do not abruptly stop taking tramadol as withdrawal symptoms like nausea, diarrhea, anxiety, sweating, difficulty in sleep, shivering, pain, tremors, or rarely, hallucinations may occur.
Consult with your doctor before discontinuing tramadol treatment; do NOT discontinue treatment on your own. Withdrawal symptoms may be relieved by re-initiation of opioid therapy followed by a slow, dose reduction combined with symptomatic support, as directed by your doctor.
Learn More: Prescription Drug Addiction: Top Facts for You & Your Family
5. What is an Opioid Analgesic REMS?
Some medications have an inherent risk that require a restricted program known as a Risk Evaluation and Mitigation Strategy (REMS) to ensure safe use. All opioid drugs like tramadol have an Opioid Analgesic REMS program in place as mandated by the FDA.
- A REMS is a medication safety program for certain medications with serious safety concerns or dangerous side effects to help ensure the benefits of the medication outweigh its risks. The FDA may require these and they are developed by the drug manufacturer.
- There are typically many steps to complete for a REMS, and can involve education and certification of healthcare providers, patients, pharmacies, and drug wholesalers.
- Creation of a Medication Guide that is updated with important patient information is part of the Opioid Analgesic REMS. The patient receives this each time they fill their opioid prescription and should review for changes.
6. Tramadol is available in both immediate-release and extended-release formulations.
Both the immediate-release and extended-release formulation of tramadol are available generically and can possibly save you hundreds of dollars on your prescription.
If you prefer generic medications due to cost-savings, ask your physician to only write for generic drugs whenever possible. If you cannot afford your medication, do not walk away from the pharmacy. Ask your doctor or pharmacist for more affordable alternatives.
The extended-release form of tramadol is for around-the-clock treatment of pain and not for use on an as-needed basis. Tramadol extended-release tablets must be taken whole, and not split, chewed or crushed.
A combination product of tramadol and acetaminophen (Ultracet) is also available by prescription in both brand and generic form.
- Combining tramadol with acetaminophen (Tylenol) may further help with pain and allow lower doses of tramadol to be used.
- However, Ultracet is not approved for use for more than 5 days.
- Also, be careful not to combine Ultracet with extra acetaminophen (Tylenol), as too much acetaminophen is toxic for your liver.
Learn more about generic drugs in “Facts About Generic Drugs”.
7. Dose adjustments are needed in the elderly, and in those with kidney or liver problems.
Here's another good reason not to share your tramadol with others: the dose you are prescribed may not be the right dose for someone else, and could lead to serious consequences like slowed or stopped breathing, coma, or even death.
As with many medications, if you are young, elderly, or have kidney or liver disease dose adjustments are often required.
- The dosing interval (how often you take the drug) may need to be adjusted, the actual dose of the drug may need to be reduced, and you may have a maximum daily dose you should not exceed.
- Talk to your doctor about the need for adjusted doses with any medication, including tramadol.
- Do not exceed the prescribed dose or dosing interval prescribed by your doctor.
Patients older than 65 years of age
- Doses should usually start at the low end of the dosing range and can be titrated upwards slowly based on tolerance and effectiveness.
Patients older than 75 years of age
- Maximum dose of regular-release tramadol oral tablets: 300 mg per day (in divided doses).
Kidney Disease and Liver Disease
Over 30% of tramadol is excreted by the kidneys as the unchanged molecule, which means it could lead to toxic blood levels in patients with kidney disease.
- Tramadol is metabolized through the liver, so a patient with kidney or liver disease may require a change in dose, different dose interval, or maximum amount of drug taken each day.
- Some patients with severe kidney or liver impairment may not be candidates for tramadol treatment at all.
- Providers should consider monitoring renal function (such as creatinine clearance) with tramadol treatment, especially in the elderly, because this drug is substantially excreted by the kidney.
8. There are ways to engage with other patients using tramadol.
Support groups may be helpful for patients who take tramadol, who use medications for pain relief, who are in need of addiction support, and for many other needs.
Joining one or more support groups is a great way to discover others taking related medications with similar medical conditions, keep up with the news, and share your own experience.
- Ultram Support Group
There are over 1300 reviews for tramadol from patients who use this drug for general pain, back pain, tooth pain and other various conditions (some of which may be off-label use, meaning the drug is not approved by the FDA for that particular use).
Remember, this information is NOT intended to endorse tramadol or recommend therapy. While these reviews may or may not be helpful to you, they are NOT a substitute for the expertise, skill, knowledge and judgement of your individual healthcare provider.
Sources
- Ultram product label. Janssen Pharmaceuticals, Inc. Revised 9/2018. Accessed Dec. 11, 2018 at https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/020281s042s043lbl.pdf
- Drug Enforcement Administration. Office of Diversion Control. Drug & Chemical Evaluation Section. Tramadol. (Trade Names: Ultram, Ultracet).
- Bush DM. Emergency Department Visits for Adverse Reactions Involving the Pain Medication Tramadol. 2015 May 14. In: The CBHSQ Report. Rockville (MD): Substance Abuse and Mental Health Services Administration (US); 2013. Accessed Dec. 11, 2018 at https://misuse.ncbi.nlm.nih.gov/error/abuse.shtml
- Drug Enforcement Administration. Office of Diversion Control. Drug and Chemical Evaluation Section. Tramadol.
(Trade Names: Ultram, Ultracet). Accessed Feb. 27, 2018. - Sansone RA, Sansone LA. Tramadol: Seizures, Serotonin Syndrome, and Coadministered Antidepressants. Psychiatry (Edgmont). 2009;6:17-21. Accessed Feb. 27, 2018 at https://misuse.ncbi.nlm.nih.gov/error/abuse.shtml
- Tramadol Product Label. Drugs.com. Accessed Dec. 12, 2018 at https://www.drugs.com/tramadol.html
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
RESOURCES > Federal Register Notices > Rules - 2014 > Final Rule: Placement of Tramadol Into Schedule IV
[Federal Register Volume 79, Number 127 (Wednesday, July 2, 2014)]
[Rules and Regulations]
[Pages 37623-37630]
From the Federal Register Online via the Government Printing Office [www.gpo.gov]
[FR Doc No: 2014-15548]
DEPARTMENT OF JUSTICE
Drug Enforcement Administration
[Docket No. DEA-351]
Schedules of Controlled Substances: Placement of Tramadol Into Schedule IV
AGENCY: Drug Enforcement Administration, Department of Justice.
ACTION: Final rule.
SUMMARY: With the issuance of this final rule, the Deputy Administrator of the Drug Enforcement Administration places the substance 2- [(dimethylamino)methyl]-1-(3-methoxyphenyl)cyclohexanol (tramadol), including its salts, isomers, and salts of isomers, into schedule IV of the Controlled Substances Act. This scheduling action is pursuant to the Controlled Substances Act which requires that such actions be made on the record after opportunity for a hearing through formal rulemaking. This action imposes the regulatory controls and administrative, civil, and criminal sanctions applicable to schedule IV controlled substances on persons who handle (manufacture, distribute, dispense, import, export, engage in research, conduct instructional activities with, or possess) or propose to handle tramadol.
DATES: Effective August 18, 2014.
FOR FURTHER INFORMATION CONTACT: Erika Gehrmann, Office of Diversion Control, Drug Enforcement Administration; Mailing Address: 8701 Morrissette Drive, Springfield, Virginia 22152; Telephone: (202) 598- 6812.
SUPPLEMENTARY INFORMATION:
Legal Authority
The Drug Enforcement Administration (DEA) implements and enforces titles II and III of the Comprehensive Drug Abuse Prevention and Control Act of 1970, as amended. Titles II and III are referred to as the 'Controlled Substances Act' and the 'Controlled Substances Import and Export Act,' respectively, but they are collectively referred to as the 'Controlled Substances Act' or the 'CSA' for the purposes of this action. 21 U.S.C. 801-971. The DEA publishes the implementing regulations for these statutes in title 21 of the Code of Federal Regulations (CFR), parts 1300 to 1321. The CSA and its implementing regulations are designed to prevent, detect, and eliminate the diversion of controlled substances and listed chemicals into the illicit market while providing for the legitimate medical, scientific, research, and industrial needs of the United States. Controlled substances have the potential for abuse and dependence and are controlled to protect the public health and safety.
Under the CSA, every controlled substance is classified in one of five schedules based upon its potential for abuse, currently accepted medical use, and the degree of dependence the drug or other substance may cause. 21 U.S.C. 812. The initial schedules of controlled substances established by Congress are found at 21 U.S.C. 812(c) and the current list of scheduled substances is published at 21 CFR part 1308.
Pursuant to 21 U.S.C. 811(a)(1), the Attorney General may, by rule, 'add to such a schedule or transfer between such schedules any drug or other substance if he (A) finds that such drug or other substance has a potential for abuse, and (B) makes with respect to such drug or other substance the findings prescribed by [21 U.S.C. 812(b)] for the schedule in which such drug is to be placed * * *.' The Attorney General has delegated scheduling authority under 21 U.S.C. 811 to the Administrator of the DEA, 28 CFR 0.100, who in turn has redelegated that authority to the Deputy Administrator of the DEA, 28 CFR part 0, appendix to subpart R. The CSA provides that scheduling of any drug or other substance may be initiated by the Attorney General (1) on his own motion, (2) at the request of the Secretary of the Department of Health and Human Services (HHS),1 or (3) on the petition of any interested party. 21 U.S.C. 811(a). This action was initiated by four petitions to schedule tramadol under the CSA, and is supported by, inter alia, a recommendation from the Assistant Secretary of the HHS and an evaluation of all relevant data by the DEA. This action imposes the regulatory controls and administrative, civil, and criminal sanctions applicable to schedule IV controlled substances on persons who handle or propose to handle tramadol.2
---------------------------------------------------------------------------
1 As discussed in a memorandum of understanding entered into by the Food and Drug Administration (FDA) and the National Institute on Drug Abuse (NIDA), the FDA acts as the lead agency within the HHS in carrying out the Secretary's scheduling responsibilities under the CSA, with the concurrence of NIDA. 50 FR 9518, Mar. 8, 1985. The Secretary of the HHS has delegated to the Assistant Secretary for Health of the HHS the authority to make domestic drug scheduling recommendations. 58 FR 35460, July 1, 1993.
2 See infra note 3.
---------------------------------------------------------------------------
Background
Tramadol is a centrally acting opioid analgesic that produces its primary opioid-like action through an active metabolite, referred to as the 'M1' metabolite (O-desmethyltramadol). It was first approved for use in the United States by the U.S. Food and Drug Administration (FDA) in 1995 under the trade name ULTRAM[supreg]. Subsequently, the FDA approved for marketing generic, combination, and extended release tramadol products.
Because of its chemical structure, 2-[(dimethylamino)methyl]-1-(3- methoxyphenyl) cyclohexanol can exist as different isomeric forms. Thus, various prefixes can be associated with the name. Some examples of these prefixes include dextro, levo, d, l, R, S, cis, trans, erythro, threo, (+), (-), racemic, and may include combinations of these prefixes sometimes with numerical designations. Any such isomer is, in fact, 2-[(dimethylamino)methyl]-1-(3-methoxyphenyl)cyclohexanol. Tramadol is typically formulated as a racemic mixture identified as ()-cis-2-[(dimethylamino)methyl]-1-(3- methoxyphenyl)cyclohexanol hydrochloride.3
---------------------------------------------------------------------------
3 For simplicity, from this point forward in the document, 'tramadol' is used to refer to 2-[(dimethylamino)methyl]-1-(3- methoxyphenyl)cyclohexanol, its salts, isomers, salts of isomers, and all isomeric configurations of possible forms.
---------------------------------------------------------------------------
HHS and DEA Eight-Factor Analyses
On September 16, 2010, the Assistant Secretary of the HHS provided to the DEA a scientific and medical evaluation and scheduling recommendation entitled 'Basis for the Recommendation to Schedule Tramadol in Schedule IV of the Controlled Substances Act.' After considering the eight factors in 21
[[Page 37624]]
U.S.C. 811(c), as well as the substance's abuse potential, legitimate medical use, and dependence liability, the Assistant Secretary of the HHS recommended that tramadol be controlled in schedule IV of the CSA under 21 U.S.C. 812(b). The DEA conducted its own eight-factor analysis of tramadol pursuant to 21 U.S.C. 811(c). Both the DEA and HHS analyses are available in their entirety in the public docket for this rule (Docket No. DEA-351) at http://www.regulations.gov under 'Supporting and Related Material.'
Determination To Schedule Tramadol
After a review of the available data, including the scientific and medical evaluation and the scheduling recommendation from the HHS, the Deputy Administrator of the DEA published in the Federal Register a notice of proposed rulemaking (NPRM) entitled 'Schedules of Controlled Substances: Placement of Tramadol Into Schedule IV' which proposed to place tramadol in schedule IV of the CSA. 78 FR 65923, Nov. 4, 2013. The proposed rule provided an opportunity for interested persons to file a request for hearing in accordance with DEA regulations by December 4, 2013. No requests for such a hearing were received by the DEA. The NPRM also provided an opportunity for interested persons to submit written comments on the proposed rule on or before January 3, 2014.
Comments Received
The DEA received 27 comments on the proposed rule to schedule tramadol. Sixteen commenters expressed support for controlling tramadol as a schedule IV controlled substance, nine commenters were opposed to tramadol being placed into schedule IV of the CSA, and two commenters did not take a position.
Support of the Proposed Rule
Sixteen commenters supported controlling tramadol as a schedule IV controlled substance. Among those 16 commenters expressing support were two State Boards of Pharmacy. One veterinary distributor's association stated that it supports the DEA designating tramadol as a schedule IV controlled substance because it will enable distributors to operate with efficiency and consistency across the United States along with requiring an increased level of due diligence and monitoring. A national veterinary medical association, a national healthcare association, and a national pharmacy association were also among those who expressed support for the rule.
Several commenters supporting the rule expressed their concern regarding the abuse potential and resulting threat to public health posed by tramadol. Writing in support of scheduling tramadol, a local multi-agency prescription drug abuse task force described tramadol as a ' 'loop hole' drug which is addictive, abused, and diverted,' but which is not yet realized as such by many patients and prescribers due to its current non-controlled status. One commenter stated that given the abuse potential of tramadol (which according to the commenter is often abused in combination with other controlled substances), scheduling this drug will ensure that it is subject to the same controls as other similarly addictive controlled substances. Yet another commenter noted that although analgesics are addictive to a very small percentage of people that use them, scheduling this drug would reduce the number of emergency room visits and number of overdose deaths.
A certified pharmacy technician described her experiences of witnessing the abuse of tramadol by patients on a daily basis. She stated the stricter controlled substance laws of the State of Mississippi have seemed to lessen the abuse. A group of pharmacy students noted that tramadol, marketed as ULTRAM[supreg], is currently the only uncontrolled opioid on the market. Another commenter who supported the rule stated: 'In the field of pharmacy, some patients have expressed concern about the reclassification of tramadol, believing that new regulations could complicate or impede new and chronic patients from receiving their prescriptions.' This commenter noted that this is a common misconception since schedule IV controlled medications are in fact readily available for those with a valid prescription and the appropriate medical condition. In addition, the commenter noted that these types of prescriptions also have the added convenience of being easily transferrable between pharmacies, phoned-in by prescribers, and refilled five times over a six month period.
DEA Response: The DEA appreciates the support for the rule.
Opposition to the Proposed Rule
1. Access to Pain Medication by the Elderly
An association for consulting pharmacists stated that controlling tramadol would limit access to needed pain medications for elderly patients and opposed the proposed scheduling until a workable solution to ensure timely access for patients in long-term care facilities (LTCFs) can be reached. Specifically, the commenter expressed concern that, should tramadol become a controlled substance, LTCF nurses would no longer be able to call-in or fax a chart order directly to the pharmacy. According to the commenter, in LTCFs, prescribers must call, hand deliver, or fax controlled substance prescriptions to pharmacies, and this in turn involves LTCF employees having to track down the (often non-employee) prescriber. This practice, according to the commenter, can severely impede delivery of prescription medications to LTCF patients.
DEA Response: The processes and procedures associated with dispensing a controlled substance are not relevant factors to the determination whether a substance should be controlled or under what schedule a substance should be placed if it is controlled. See 21 U.S.C. 811 and 812. Nonetheless, controlling tramadol as a schedule IV controlled substance should not hinder legitimate access to the medicine, whether within the LTCF setting or elsewhere. As summarized by a State Board of Pharmacy who wrote in support of controlling tramadol: 'Scheduling a medication does not make it impossible to prescribe, dispense and administer the medication. However, it does alert practitioners, dispensers and perhaps even some patients that the medication has some potential dangers for addiction and misuse, and frequent monitoring and evaluation by practitioners and dispensers of such drugs is necessary for appropriate patient care.'
Currently, tramadol is a non-controlled medication that the FDA has approved only for prescription use. Tramadol, as a schedule IV controlled substance, will continue to require a prescription, either orally or in writing. 21 U.S.C. 829(b). The CSA allows for the legitimate prescribing and use of controlled substances; therefore, the control of tramadol should not hinder patient access to the medication. The prescription for tramadol, as a controlled substance, may only be issued by an individual practitioner who is either registered with the DEA or exempt from registration. 21 CFR 1306.03. A prescription for a controlled substance must also be issued for a legitimate medical purpose by an individual practitioner acting in the course of his professional practice. 21 CFR 1306.04(a). Upon the effective date of this rule, tramadol prescriptions may be filled up to six months after the date prescribed, and may be refilled up to
[[Page 37625]]
five times within six months after the date on which such prescription was issued. 21 U.S.C. 829(b); 21 CFR 1306.22 (a) and (e); see also 21 CFR 1306.23 (b) and (c). In addition, there are no dosage unit limitations for prescriptions for schedule III, IV, or V controlled substances unless the controlled substance is prescribed for administration to an ultimate user who is institutionalized. 21 CFR 1306.24(c).
The substantive requirement that a practitioner acting in the usual course of professional practice determine that tramadol is medically necessary to treat the patient does not hinder legitimate access; the procedural requirements relating to transmission of a legitimate prescription do not hinder legitimate access either. Once an individual practitioner makes a medical determination to prescribe a schedule III through V controlled substance, a prescriber's agent may call-in or fax a prescription for it. See 21 CFR 1306.03(b), 1306.21(a). The DEA recognizes the unique challenges pertaining to handling and using controlled substances at LTCFs and has previously addressed related concerns.4 A DEA registered practitioner may not delegate to a nurse, a pharmacist, or anyone else his or her authority to make a medical determination whether to prescribe a particular controlled substance. However, oral prescriptions for controlled substances in schedules III- V may be communicated to a pharmacy by an employee or agent of the prescribing practitioner, 21 CFR 1306.03(b). Note that the prescribing practitioner remains responsible for ensuring that the prescription conforms 'in all essential respects to the law and regulations,' 21 CFR 1306.05(f). 75 FR 61613, 61614, Oct. 6, 2010. This requires the practitioner alone to determine--on a prescription by prescription basis--whether the prescription is supported by a legitimate medical purpose and that all the essential elements of the prescription are met.
---------------------------------------------------------------------------
4 E.g., 'Preventing the Accumulation of Surplus Controlled Substances at Long Term Care Facilities,' 66 FR 20833, Apr. 25, 2001; 'Role of Authorized Agents in Communicating Controlled Substance Prescriptions to Pharmacies,' 75 FR 61613, Oct. 6, 2010.
---------------------------------------------------------------------------
2. Fear of Criminal Action
Some commenters expressed concern that scheduling tramadol would deter prescribers from properly treating pain for fear of facing criminal action.
DEA Response: One of the most important principles underlying the CSA is that every prescription for a controlled substance must be issued for a legitimate medical purpose by an individual practitioner acting in the usual course of his professional practice. 21 CFR 1306.04(a); U.S. v. Moore, 423 U.S. 122 (1975) (holding registered physicians may be prosecuted for violation of the CSA when their activities fall outside the usual course of professional practice). The DEA Policy Statement entitled 'Dispensing Controlled Substances for the Treatment of Pain,' 71 FR 52715 (Sept. 6, 2006), makes clear that this longstanding requirement should in no way interfere with the legitimate practice of medicine or cause any practitioner to be reluctant to provide legitimate pain treatment. Providers (as well as ultimate users) become subject to administrative, civil, and/or criminal proceedings when their activity involving controlled substances is not authorized by, or in violation of, the CSA.
3. Shift to the Black-Market
Several commenters stated that scheduling tramadol would limit their access to tramadol, causing them to have to buy tramadol on the street.
DEA Response: As discussed above, schedule IV controlled medications are readily available for legitimate medical use.
4. Scientific Data Not Sufficient
One commenter reviewed selected published literature and submitted a short review document with a conclusion that 'the current available scientific evidence supports the continuation of a non-controlled classification' of tramadol.
DEA Response: The CSA mandates that both the HHS and DEA conduct a review of the drug or other substance as related to the eight factors enumerated in 21 U.S.C. 811(c): (1) Its actual or relative potential for abuse; (2) scientific evidence of its pharmacological effect, if known; (3) the state of current scientific knowledge regarding the drug or other substance; (4) its history and current pattern of abuse; (5) the scope, duration, and significant of abuse; (6) what, if any, risk there is to the public health; (7) its psychic or physiological dependence liability; and (8) whether the substance is an immediate precursor of a substance already controlled. The Assistant Secretary of the HHS provided a scientific and medical evaluation and a scheduling recommendation to control tramadol as a schedule IV controlled substance. In accordance with 21 U.S.C. 811(c), the DEA conducted its own analysis of the eight factors determinative of control. Besides published literature, various other data as detailed in the supporting documents were considered in making the scheduling determination for tramadol. Thus, the scheduling determination is based on a comprehensive evaluation of all available data as related to the above mentioned eight factors. The summary of each factor as analyzed by the HHS and the DEA, and as considered by the DEA in this scheduling action, was provided in the proposed rule. Both the DEA and the HHS analyses have been made available in their entirety under 'Supporting and Related Material' of the public docket for this rule at http://www.regulations.gov under Docket No. DEA-351. As discussed in detail in the DEA's eight-factor analysis, collectively, the available information regarding tramadol supports an abuse potential that is less than that of schedule III and similar to that for schedule IV. Preclinical self-administration studies show that tramadol produces limited reinforcing effects, consistent with schedule IV. At supra-therapeutic doses, tramadol can produce subjective reinforcing effects similar to that of morphine (C-II) and approaching that of oxycodone (C-II). At high doses (but not therapeutic doses), tramadol can produce subjective reinforcing effects similar to propoxyphene (C-IV). For both tramadol and propoxyphene, the doses required to produce significant subjective reinforcing effects are in a range causing sufficient adverse effects. These observations indicate that the subjective reinforcing effects, a reflection of abuse potential, of tramadol are less than that of morphine or oxycodone, but similar to that of propoxyphene.
Based on the review of the HHS evaluation and scheduling recommendation and all other relevant data, the DEA has found that tramadol has an abuse potential and meets the requirements for schedule IV controls under the CSA.
5. Disagreement With Tramadol Classification as an Opioid
One commenter who supported the rule stated that tramadol should not be compared to hydrocodone because hydrocodone is an opioid and tramadol is psychotropic in nature and very similar to, if not the same as, a serotonin-norepinephrine reuptake inhibitor (SNRI).
DEA Response: In the NPRM and supporting documents, the DEA compared tramadol mainly to propoxyphene (narcotic schedule IV). Based on both the HHS and the DEA analyses, there is strong scientific evidence that tramadol and propoxyphene are similar regarding
[[Page 37626]]
their behavioral pharmacology and abuse potential pattern, thus suggesting that it is appropriate to control tramadol as a schedule IV controlled substance.
In addition, as stated in the supporting scientific documents, both the HHS and the DEA deem tramadol to be an opioid because tramadol shares similar pharmacological activities with opioids that are controlled under the CSA (schedules II-IV). (The labeling for FDA approved tramadol products states that tramadol is a centrally acting opioid analgesic.) An examination of the general pharmacology (including behavioral pharmacology) of tramadol reveals that tramadol produces many pharmacological effects similar to those of other opioids. These pharmacological effects include, but are not limited to, analgesia, respiratory depression, miosis, cough suppression, and inhibition of bowel mobility, and as such, tramadol is considered an opioid. The opioid pharmacology of tramadol primarily resides with its metabolite, O-desmethyltramadol, designated 'M1,' and to a much lesser extent with tramadol, the parent drug. In addition, tramadol resembles some opioids insofar as it has the additional pharmacological effects of blocking the reuptake of norepinephrine and serotonin.
The CSA defines an 'opiate' as 'any drug or other substance having an addiction-forming or addiction-sustaining liability similar to morphine or being capable of conversion into a drug having such addiction-forming or addiction-sustaining liability.' 21 U.S.C. 802(18). Opium, opiates, derivatives of opium and opiates, including their isomers, whether produced directly or indirectly by extraction from substances of vegetable origin, or independently by means of chemical synthesis, are 'narcotic drugs' as defined by the CSA, 21 U.S.C. 802(17).5 As discussed in the supporting eight-factor documentation, preclinical studies demonstrate that tramadol, as other opioids in schedules I through IV, exhibits complete generalization to morphine and is able to produce some reinforcing effects. Repeated administration of tramadol in animals caused dependence development, evidenced by a withdrawal syndrome similar in intensity to pentazocine (schedule IV) or propoxyphene (narcotic schedule IV).
---------------------------------------------------------------------------
5 Including their isomers, esters, ethers, salts, and salts of isomers, whenever the existence of such isomers, esters, ethers, and salts is possible within the specific chemical designation; however, does not include the isoquinoline alkaloids of opium.
---------------------------------------------------------------------------
Although, generally, the controls imposed by the CSA on drugs and other substances depend on the schedule into which they are placed, there are certain additional requirements and restrictions for narcotic drugs. For example, narcotic drugs in schedule III, IV, or V may not be imported into the United States unless it is found that such importation is needed to provide for the legitimate medical, scientific, or other legitimate purposes under the specified, limited circumstances described in 21 U.S.C. 952(a). Narcotic controlled substances may not be exported unless the conditions imposed by 21 U.S.C. 953(a) are satisfied.
6. Never-Ending Practice of Drug Scheduling
Two commenters raised concerns that, despite the scheduling of drugs such as tramadol, individuals will always find substances to abuse, thus creating 'a never ending story of scheduling drugs.'
DEA Response: Pursuant to 21 U.S.C. 811(a), the CSA authorizes the DEA, under authority delegated by the Attorney General, to add to such a schedule any drug or other substance if it is found that the drug or other substance has a potential for abuse, and makes with respect to such drug or other substance the findings prescribed by 21 U.S.C. 812(b). As such, the scheduling authority established by Congress specifically allows new substances to be added to the list of controlled substances without regard to the number of substances already controlled. See also 21 U.S.C. 812(a) ('Such schedules shall initially consist of * * *' (emphasis added)).
Requests for Staggered Implementation of Various Portions of the Rule
A national association that represents primary healthcare distributors commented that although they recognized the underlying reasons for scheduling tramadol and agreed with the reasoning and basis for controlling tramadol, the DEA should provide an extended time period before implementation to allow registrants to become compliant with portions of the rule regarding security, labeling and packaging, and reporting.6 The association requested that the requirement for conducting inventory of tramadol products within wholesale distribution centers take place as of the effective date of the final scheduling decision. The association's concerns (as well as the DEA's responses) are outlined and discussed below.
---------------------------------------------------------------------------
6 Pursuant to 5 U.S.C. 553(d) and in accordance with 21 CFR 1308.45, a final rule scheduling a substance shall not be effective less than 30 days from the date of its publication in the Federal Register unless the Administrator finds that conditions of public health or safety necessitate an earlier effective date.
---------------------------------------------------------------------------
Schedule 2 Medications
1. Request for Staggered Effective Dates, Generally
The association requested that the DEA implement handling requirements for tramadol in stages. For example, they requested that the requirement for conducting inventory of tramadol products within wholesale distribution centers take place as of the effective date of the final scheduling decision but delaying the requirements for compliance with the security provisions of 21 CFR 1301.71-1301.93.
DEA Response: Generally, scheduling actions for drugs and other substances currently marketed in the United States are effective 30 days from the date of publication of the final rule in the Federal Register. In order to ensure the continued availability of tramadol for legitimate medical use, while also ensuring it is not subject to misuse, abuse, and diversion, the DEA is establishing an effective date of this final rule for all handling requirements 45 days from the date of publication. This 45-day period will provide a reasonable time for registrants to comply with the handling requirements for a schedule IV controlled substance and was established upon a full consideration of the totality of circumstances specific to tramadol.
Although the DEA has in the past, for some scheduling actions, allowed for additional time for compliance with certain handling requirements beyond the general effective date, the DEA has specifically chosen to forgo staggered implementation dates of handling requirements as different implementation dates leads to confusion and inconsistent application of the law.
2. Security
The association recommended a minimum of 120 days from the date of the final rule to allow for compliance in order to provide storage, revise operating procedures, train staff, and amend monitoring systems.
DEA Response: In order to ensure the continued availability of tramadol for legitimate medical use, while also ensuring it is not subject to misuse, abuse, and diversion, the DEA is establishing an effective date of this final rule, including security requirements, 45 days from the date of publication. Upon promulgation, registrants must comply with the applicable security provisions of 21 CFR 1301.71- 1301.93. This 45-day period will provide a reasonable time for registrants to comply with the security
[[Page 37627]]
requirements for a schedule IV controlled substance. As noted by the association, it is believed that distributors of tramadol already have adequate space within their warehouse cages to store the anticipated volume of tramadol and 'thus construction or expansion of cage space is unlikely to result * * *.' Accordingly, it is reasonably likely that handlers and proposed handlers of tramadol have already instituted or made plans to institute the necessary modifications regarding security, including amendments to their suspicious orders monitoring systems to include tramadol orders. In order to provide handlers of tramadol a reasonable time period to comply with schedule IV handling requirements, including those for security, the DEA is allowing an additional 15 days, as compared to the generally allotted 30 days, from publication in the Federal Register before this rule becomes effective. After 45 days from the date of the final rule, tramadol will be subject to schedule III-V security requirements.
The DEA has carefully considered the security requirements for compliance with this rule. As confirmed by the association, current distributors of tramadol are DEA registrants with existing controlled substance storage that complies with DEA regulations. The DEA understands that handlers of tramadol may need to make modifications to their current security procedures for compliance. These modifications necessary for security compliance will be a one-time modification to provide for the appropriate storage, revision of operating procedures, training of staff, and amendments to suspicious order monitoring systems to include customer verifications. The DEA believes that a 45- day period will provide handlers of tramadol adequate time to implement these one-time modifications in compliance with the DEA security regulations. Registrants are familiar with the applicable security regulations, and already have systems in place with respect to other controlled substances. Accordingly, revising operating procedures, amending monitoring systems, and training staff with respect to tramadol should be easily accomplished within the 45-day compliance timeframe. The DEA strongly advises current registrants (and those entities that may seek registration as a result of this action) to work closely with their local DEA office regarding the applicable security requirements and any necessary modifications due to compliance with this rule. 21 CFR 1301.71(d).
3. Distribution of Products With the Pre-Control Label
The association stated that in accordance with 21 CFR 1302.05, the DEA has the authority to set a date on which labeling and packaging requirements will become effective, and requested clarification of when the distribution of products with the pre-scheduling label should cease. The association also requested clarification as to whether the cessation of the manufacture of products for commercial containers with the pre-scheduling labeling will also mean that manufacturers would be required to cease distribution to wholesale distributors of products they might have in stock bearing the pre-scheduling label. The association stated that the ambiguity of the compliance period poses a dilemma for those in the tramadol supply chain, and requested the DEA to act to meet healthcare needs and avoid waste by allowing products bearing the pre-scheduling label to move through the supply chain until the inventory is depleted. Alternatively, the association suggested that the DEA allow distributors to continue to sell pre-scheduling labeled product for at least 180 days after the effective date of the final rule.
DEA Response: As of the effective date of the final rule, pursuant to 21 U.S.C. 821, 825, and 958(e) and in accordance with 21 CFR 1302.03, manufacturers are required to print upon the labeling of each commercial container of tramadol they distribute the designation of tramadol as 'C-IV.' It shall be unlawful for commercial containers of tramadol to be distributed without bearing the label properly identifying it as a schedule IV controlled substance in accordance with 21 CFR part 1302. As clearly stated in 21 CFR 1302.05, '[a]ll labels on commercial containers of, and all labeling of, a controlled substance which either is transferred to another schedule or is added to any schedule shall comply with the requirements of Sec. 1302.03, on or before the effective date established in the final order for the transfer or addition.' Accordingly, the DEA is requiring that commercial containers of tramadol distributed on or after 45 days from the date of publication of the final rule be labeled as 'C-IV' and be packaged in accordance with 21 CFR part 1302.
From the 2007 Economic Census, the DEA estimates that the inventory turnover ratio for the industry 7 is approximately 11.3.8 The inventory turnover ratio represents the number of times the inventory sells (turns) in a year. The 11.3 inventory turnover ratio equates to an average of 32 days to sell inventory. The 11.3 turnover ratio is consistent with that of large distributors where financial information was publicly available and reviewed. Publicly reviewed data reports that about 85% of all revenues (an indirect indicator of dosage units moved) from drug distribution in the United States come from three public wholesalers, each with annual revenue in the billions. The DEA additionally notes that many regional and specialist pharmaceutical wholesalers have been acquired by the largest three distribution companies. The inventory turnover ratio is a reasonable estimate for the entire industry and all products under the circumstances. Because the 32 days to sell inventory is an average based on industry-wide census data, it is possible for an individual company and/or product line to have shorter or longer time to sell.
---------------------------------------------------------------------------
7 NAICS 424210--Drugs and druggists' sundries merchant wholesalers; Merchant wholesalers, except manufacturers' sales branches and offices.
8 The inventory turnover ratio of 11.3 was calculated by dividing the 2007 'cost of goods sold' for the industry of $280,481,051,000 by the average end-of-year 2006 total inventories of $24,782,835,000.
---------------------------------------------------------------------------
Since tramadol is a widely prescribed drug, with nearly 40 million prescriptions written in 2012,9 the DEA expects distributors to receive and distribute tramadol at high volume and with regularity; thus, anticipating shorter than average days to sell tramadol than overall industry average inventory. However, to accommodate those distributors that have lower than average industry turnover ratio, the DEA is establishing an effective date of this final rule, including labeling and packaging requirements, 45 days from the date of publication. The DEA believes this will provide a reasonable time for distributors to sell existing stock with pre-control labeling and packaging and to stock inventory with post-control labeling and packaging.
---------------------------------------------------------------------------
9 IMS Health, National Sales PerspectiveTM (NSP).
---------------------------------------------------------------------------
Additionally, the DEA believes that any distributor that requires more than 45 days to sell tramadol inventory under normal circumstances can make minor modifications to ordering and stocking procedure for a transitional period to meet the established effective date at minimal cost. Distributors also have the option of returning excess stock of tramadol product without the 'C-IV'
[[Page 37628]]
label to the manufacturer, as authorized by 21 CFR 1307.12. The DEA takes this opportunity to clarify that the regulation pertaining to labeling of commercial containers applies only to distributions by manufacturers and distributors. The DEA does not regulate the labeling and packing of commercial containers of controlled substances downstream of distributors.
As summarized in the NPRM, and discussed in detail in the supporting eight factor analyses, tramadol meets the statutory requirements for control and for placement in schedule IV. Based upon the reasons discussed above, the DEA believes that 45 days is a reasonable amount of time for registrants to modify their operations so that the necessary safeguards are in place to prevent the abuse and diversion of tramadol.
4. Automation of Reports and Consolidated Orders System ('ARCOS') Reporting
The association stated that only schedule I and II (and some schedule III) products are subject to reporting under the DEA's Automation of Reports and Consolidated Orders System ('ARCOS'), so it would be an error to require distributors to report tramadol (a schedule IV narcotic) to ARCOS.
DEA Response: DEA regulations do not require distributors to file ARCOS reports for schedule IV narcotics.
Scheduling Conclusion
Based on consideration of all comments, the scientific and medical evaluation and accompanying recommendation of the HHS, and based on the DEA's consideration of its own eight-factor analysis, the DEA finds that these facts and all other relevant data constitute substantial evidence of potential for abuse of tramadol. As such, the DEA is scheduling tramadol as a controlled substance under the CSA.
Determination of Appropriate Schedule
The CSA establishes five schedules of controlled substances known as schedules I, II, III, IV, and V. The CSA outlines the findings required for placing a drug or other substance in any particular schedule. 21 U.S.C. 812(b). After consideration of the analysis and recommendation of the Assistant Secretary for Health of the HHS and review of all relevant and available data, the Deputy Administrator of the DEA, pursuant to 21 U.S.C. 812(b)(4), finds that:
1. Tramadol has a low potential for abuse relative to the drugs or substances in schedule III. The abuse potential of tramadol is comparable to the schedule IV controlled substance propoxyphene;
2. Tramadol has a currently accepted medical use in treatment in the United States. Tramadol and other tramadol-containing products are approved for marketing by the FDA to manage moderate to moderately severe pain; and
3. Abuse of tramadol may lead to limited physical dependence or psychological dependence relative to the drugs or other substances in schedule III.
Based on these findings, the Deputy Administrator of the DEA concludes that tramadol, including its salts, isomers, and salts of isomers, warrants control in schedule IV of the CSA. 21 U.S.C. 812(b)(4).
Requirements for Handling Tramadol
Adobe services are available only to users 13 and older and require agreement to additional terms of use and the Adobe Privacy Policy. Applications and services may not be available in all countries or languages and may be subject to change or discontinuation without notice. This product may integrate with or allow access to certain Adobe or third-party hosted online services. Adobe photoshop lightroom 6 torrent.
Upon the effective date of this final rule, any person who handles tramadol is subject to the CSA's schedule IV regulatory controls and administrative, civil, and criminal sanctions applicable to the manufacture, distribution, dispensing, importing, exporting, engagement in research, and conduct of instructional activities, of schedule IV controlled substances including the following:
Registration. Any person who handles (manufactures, distributes, dispenses, imports, exports, engages in research, or conducts instructional activities with) tramadol, or who desires to handle tramadol, must be registered with the DEA to conduct such activities, pursuant to 21 U.S.C. 822, 823, 957, and 958, and in accordance with 21 CFR parts 1301 and 1312 as of August 18, 2014. Any person who currently handles tramadol and is not registered with the DEA must submit an application for registration and may not continue to handle tramadol as of August 18, 2014 unless the DEA has approved that application, pursuant to 21 U.S.C. 822, 823, 957, and 958, and in accordance with 21 CFR parts 1301 and 1312.
Disposal of stocks. Any person who does not desire or is not able to obtain a schedule IV registration must surrender all quantities of currently held tramadol in accordance with the procedures outlined in 21 CFR 1307.21 on or before August 18, 2014, or may transfer all quantities of currently held tramadol to a person registered with the DEA on or before August 18, 2014.
Security. Tramadol is subject to schedule III-V security requirements and must be handled and stored pursuant to 21 U.S.C. 821 and 823, and in accordance with 21 CFR 1301.71-1301.93 as of August 18, 2014.
Labeling and Packaging. All labels and labeling for commercial containers of tramadol must comply with 21 U.S.C. 825 and 958(e), and be in accordance with 21 CFR part 1302 as of August 18, 2014.
Inventory. Every DEA registrant who possesses any quantity of tramadol on the effective date of this final rule must take an inventory of all stocks of tramadol on hand as of August 18, 2014, pursuant to 21 U.S.C. 827 and 958, and in accordance with 21 CFR 1304.03, 1304.04, and 1304.11 (a) and (d).
Any person who becomes registered with the DEA after August 18, 2014 must take an initial inventory of all stocks of controlled substances (including tramadol) on hand on the date the registrant first engages in the handling of controlled substances, pursuant to 21 U.S.C. 827 and 958, and in accordance with 21 CFR 1304.03, 1304.04, and 1304.11 (a) and (b).
After the initial inventory, every DEA registrant must take a new inventory of all stocks of controlled substances (including tramadol) on hand every two years, pursuant to 21 U.S.C. 827 and 958, and in accordance with 21 CFR 1304.03, 1304.04, and 1304.11.
Records and Reports. All DEA registrants must maintain records with respect to tramadol pursuant to 21 U.S.C. 827 and 958 and in accordance with 21 CFR parts 1304 and 1312 as of August 18, 2014.
Prescriptions. All prescriptions for tramadol or products containing tramadol must comply with 21 U.S.C. 829, and be issued in accordance with 21 CFR part 1306 and subpart C of 21 CFR part 1311 as of August 18, 2014.
Importation and Exportation. All importation and exportation of tramadol must be in compliance with 21 U.S.C. 952, 953, 957, and 958, and be in accordance with 21 CFR part 1312 as of August 18, 2014.
Liability. Any activity involving tramadol not authorized by, or in violation of, the CSA, occurring as of August 18, 2014 is unlawful, and may subject the person to administrative, civil, and/or criminal action.
Regulatory Analyses
Executive Orders 12866 and 13563
In accordance with 21 U.S.C. 811(a), this scheduling action is subject to formal rulemaking procedures done 'on the record after opportunity for a hearing,' which are conducted pursuant to the provisions of 5 U.S.C. 556 and 557. The CSA sets forth the criteria for scheduling a drug or other substance. Such actions are exempt from review by the Office of Management and Budget
[[Page 37629]]
(OMB) pursuant to section 3(d)(1) of Executive Order 12866 and the principles reaffirmed in Executive Order 13563.
Executive Order 12988
This regulation meets the applicable standards set forth in sections 3(a) and 3(b)(2) of Executive Order 12988 Civil Justice Reform to eliminate drafting errors and ambiguity, minimize litigation, provide a clear legal standard for affected conduct, and promote simplification and burden reduction.
Executive Order 13132
This rulemaking does not have federalism implications warranting the application of Executive Order 13132. The rule does not have substantial direct effects on the States, on the relationship between the national government and the States, or the distribution of power and responsibilities among the various levels of government. Resident evil streaming.
Executive Order 13175
This rule does not have tribal implications warranting the application of Executive Order 13175. This rule does not have substantial direct effects on one or more Indian tribes, on the relationship between the Federal Government and Indian tribes, or on the distribution of power and responsibilities between the Federal Government and Indian tribes.
Regulatory Flexibility Act
The Deputy Administrator, in accordance with the Regulatory Flexibility Act (RFA) (5 U.S.C. 601-612), has reviewed this final rule and by approving it certifies that it will not have a significant economic impact on a substantial number of small entities. The purpose of this final rule is to place tramadol, including its salts, isomers, and salts of isomers, into schedule IV of the CSA. By this final rule, tramadol will remain in schedule IV unless and until additional scheduling action is taken to either transfer it between the schedules or to remove it from the list of schedules. See 21 U.S.C. 811 and 812. No less restrictive measures (i.e., non-control or control in schedule V) enable the DEA to meet its statutory obligations under the CSA.
This rule affects approximately 1.5 million DEA registrations, representing approximately 376,904 entities. The DEA estimates that 367,046 (97%) of these entities are 'small entities' in accordance with the RFA and SBA size standards. 5 U.S.C. 601(6) and 15 U.S.C. 632.
In accordance with the RFA, the DEA evaluated the impact of this rule on small entities. Specifically, the DEA examined the registration, storage, inventory and recordkeeping, and disposal requirements for the 367,046 small entities estimated to be affected by the rule: 55 manufacturers; 1,418 distributors/importers/exporters; 50,032 pharmacies; and 315,541 entities employing or holding registrations as individual practitioners/mid-level practitioners/ hospitals/clinics. Ten States currently control tramadol as a schedule IV controlled substance under State law, with requirements that meet or exceed the DEA's requirements for schedule IV controlled substances discussed in the NPRM. Entities in these States are not economically impacted by this rule.
Based on the DEA's understanding of its registrants' operations and facilities, the DEA estimates a non-recurring expense for system modification and initial inventory cost of $245.01 for all entities and an additional $10,000 for secure storage for 50% of distributors, importers, and exporters. As discussed in the EIA prepared in association with the development of this final rule, manufacturers, pharmacies, physician offices/hospitals/clinics/other health care facilities, and 50% of distributors, importers, and exporters are assumed to meet the requirement of the rule without the need to expand secure storage area. The DEA estimates these costs, on an annualized basis, will have significant economic impact (cost greater than 1% of annual revenue) on 0 of 55 (0%) of small manufacturers; 50 of 1,418 (3.5%) of small distributors; 107 of 50,032 (0.2%) small business pharmacies; and 661 of 315,541 (0.2%) of individual practitioners/mid- level practitioners/hospitals/clinics, totaling 818 of 367,046 (0.2%) of all small entities. The percentage of small entities with significant economic impact is not substantial, and therefore, this rule will not result in significant economic impact on a substantial number of small entities.
Unfunded Mandates Reform Act of 1995
In accordance with the Unfunded Mandates Reform Act (UMRA) of 1995 (2 U.S.C. 1501 et seq.), the DEA has determined and certifies pursuant to UMRA that this action would not result in any Federal mandate that may result 'in the expenditure by State, local, and tribal governments, in the aggregate, or by the private sector, of $100 million or more (adjusted for inflation) in any one year * * *.' Therefore, neither a Small Government Agency Plan nor any other action is required under provisions of UMRA of 1995.
Paperwork Reduction Act of 1995
This action does not impose a new collection of information requirement under the Paperwork Reduction Act of 1995 (44 U.S.C. 3501- 3521). This action would not impose recordkeeping or reporting requirements on State or local governments, individuals, businesses, or organizations. An agency may not conduct or sponsor, and a person is not required to respond to, a collection of information unless it displays a currently valid OMB control number.
Congressional Review Act
This rule is not a major rule as defined by section 804 of the Small Business Regulatory Enforcement Fairness Act of 1996 (Congressional Review Act (CRA)). This rule will not result in: an annual effect on the economy of $100 million or more; a major increase in costs or prices for consumers, individual industries, Federal, State, or local government agencies, or geographic regions; or significant adverse effects on competition, employment, investment, productivity, innovation, or on the ability of United States-based companies to compete with foreign-based companies in domestic and export markets. However, pursuant to the CRA, the DEA has submitted a copy of this final rule to both Houses of Congress and to the Comptroller General.
List of Subjects in 21 CFR Part 1308
Administrative practice and procedure, Drug traffic control, Reporting and recordkeeping requirements.
For the reasons set out above, 21 CFR part 1308 is amended as follows:
PART 1308--SCHEDULES OF CONTROLLED SUBSTANCES
- 1. The authority citation for 21 CFR part 1308 continues to read as follows:
Authority:21 U.S.C. 811, 812, 871(b), unless otherwise noted.
- 2. Amend Sec. 1308.14 by adding a new paragraph (b)(3) to read as follows:
Sec. 1308.14 Schedule IV.
* * * * *
(b) * * *
(3) 2-[(dimethylamino)methyl]-1-(3-methoxyphenyl)cyclohexanol, its salts, optical and geometric isomers and salts of these isomers (including tramadol)—9752
* * * * *
[[Page 37630]]
Dated: June 27, 2014.
Thomas M. Harrigan,
Deputy Administrator.
[FR Doc. 2014-15548 Filed 7-1-14; 8:45 am]
BILLING CODE 4410-09-P
NOTICE: This is an unofficial version. An official version of this publication may be obtained directly from the Government Publishing Office (GPO).
| Clinical data | |
|---|---|
| Pronunciation | tra' ma dole |
| Trade names | Ultram, Zytram, others[2] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695011 |
| License data | |
| Pregnancy category |
|
| Dependence liability | Present[1] |
| Routes of administration | By mouth, IV, IM, rectal |
| Drug class | Opiate analgesic[3] |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 70–75% (by mouth), 77% (rectal), 100% (IM)[4] |
| Protein binding | 20%[1] |
| Metabolism | Liver-mediated demethylation and glucuronidation via CYP2D6 & CYP3A4[4][5] |
| Metabolites | O-desmethyltramadol, N-desmethryltramadol |
| Onset of action | Less than 1 hour (by mouth)[1] |
| Elimination half-life | 6.3 ± 1.4 h[5] |
| Duration of action | 6 hours[6] |
| Excretion | Urine (95%)[7] |
| Identifiers | |
| |
| CAS Number |
|
| PubChemCID | |
| DrugBank |
|
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI |
|
| ChEMBL | |
| CompTox Dashboard(EPA) | |
| ECHA InfoCard | |
| Chemical and physical data | |
| Formula | C16H25NO2 |
| Molar mass | 263.381 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 180 to 181 °C (356 to 358 °F) |
| |
| |
| (what is this?)(verify) | |
Tramadol, sold under the brand name Ultram among others,[2] is an opioidpain medication used to treat moderate to moderately severe pain.[1] When taken by mouth in an immediate-release formulation, the onset of pain relief usually begins within an hour.[1] It is also available by injection.[8] It may be sold in combination with paracetamol (acetaminophen) or as longer-acting formulations.[1][8]
Common side effects include constipation, itchiness, and nausea.[1] Serious side effects may include seizures, increased risk of serotonin syndrome, decreased alertness, and drug addiction.[1] A change in dosage may be recommended in those with kidney or liver problems.[1] It is not recommended in those who are at risk of suicide or in those who are pregnant.[1][8] While not recommended in women who are breastfeeding, those who take a single dose should not generally stop breastfeeding.[9]
Tramadol acts by binding to μ-opioid receptors on neurons.[1][10] It is also a serotonin–norepinephrine reuptake inhibitor (SNRI).[1][10] It is converted in the liver to O-desmethyltramadol, an opioid with stronger binding to the μ-opioid receptor.[1][11]
Tramadol was patented in 1963 and launched under the name 'Tramal' in 1977 by the West German pharmaceutical companyGrünenthal GmbH.[10][12] In the mid-1990s, it was approved in the United Kingdom and the United States.[10] It is available as a generic medication and marketed under many brand names worldwide.[1][2] In the United States, the wholesale cost is less than US$0.05 per dose as of 2018.[13] In 2016, it was the 39th most prescribed medication in the United States, with more than 19 million prescriptions.[14]
- 1Medical uses
- 2Contraindications
- 3Side effects
- 6Pharmacology
- 6.2Mechanism of action
- 7Chemistry
- 8Society and culture
- 9Research
Medical uses[edit]
Tramadol is used primarily to treat mild to severe pain, both acute and chronic.[15][16]
Its analgesic effects take about one hour to come into effect and 2 to 4 h to peak after oral administration with an immediate-release formulation.[16][15] On a dose-by-dose basis, tramadol has about one-tenth the potency of morphine and is practically equally potent when compared with pethidine and codeine.[17]
For pain moderate in severity, its effectiveness is equivalent to that of morphine; for severe pain it is less effective than morphine.[15] These painkilling effects last about 6 h.[16]
Available dosage forms include liquids, syrups, drops, elixirs, effervescent tablets and powders for mixing with water, capsules, tablets including extended-release formulations, suppositories, compounding powder, and injections.[15]
Fibromyalgia[edit]
As of 2015, tramadol was not approved in the United States for fibromyalgia.[18] Based on three small trials with weak study design, fair evidence was found for tramadol as a second-line treatment.[18]
Contraindications[edit]
Use of tramadol is not advised for people deficient in CYP2D6 enzymes.[7] The enzymes are crucial to the therapeutic effects of tramadol, by means of enabling tramadol's metabolism to desmetramadol.[15]
Pregnancy and lactation[edit]
Tramadol's use in pregnancy is generally avoided, as it may cause some reversible withdrawal effects in the newborn.[19] A small prospective study in France found, while an increased risk of miscarriages existed, no major malformations were reported in the newborn.[19] Its use during lactation is also generally advised against, but a small trial found that infants breastfed by mothers taking tramadol were exposed to about 2.88% of the dose the mothers were taking. No evidence of this dose having a harmful effect on the newborn was seen.[19]
Labour and delivery[edit]
Its use as an analgesic during labour is generally advised against due to its long onset of action (1 hour).[19] The ratio of the mean concentration of the drug in the fetus compared to that of the mother when it is given intramuscularly for labour pains has been estimated to be 1:94.[19]
Children[edit]
Its use in children is generally advised against, although it may be done under the supervision of a specialist.[15] On September 21, 2015, the FDA started investigating the safety of tramadol in use in persons under the age of 17. The investigation was initiated because some of these people have experienced slowed or difficult breathing.[20] The FDA lists age under 12 years old as a contraindication.[21][22]
Elderly[edit]
The risk of opioid-related adverse effects such as respiratory depression, falls, cognitive impairment and sedation is increased.[15]
Liver and kidney failure[edit]
The drug should be used with caution in those with liver or kidney failure, due to metabolism in the liver (to desmetramadol) and elimination by the kidneys.[15]
Side effects[edit]
The most common adverse effects of tramadol include nausea, dizziness, dry mouth, indigestion, abdominal pain, vertigo, vomiting, constipation, drowsiness, and headache.[24][25] Compared to other opioids, respiratory depression and constipation are considered less of a problem with tramadol.[25]
Chronic opioid administration may induce a state of immune tolerance,[26] although in contrast to typical opioids, it may enhance immune function.[27][28][29]
Dependence and withdrawal[edit]
Long-term use of high doses of tramadol causes physical dependence and withdrawal syndrome.[30] These include both symptoms typical of opioid withdrawal and those associated with serotonin–norepinephrine reuptake inhibitor withdrawal; symptoms include numbness, tingling, paresthesia, and tinnitus.[31] Psychiatric symptoms may include hallucinations, paranoia, extreme anxiety, panic attacks, and confusion.[32] In most cases, tramadol withdrawal will set in 12–20 hours after the last dose, but this can vary.[31] Tramadol withdrawal typically lasts longer than that of other opioids. Seven days or more of acute withdrawal symptoms can occur as opposed to typically 3 or 4 days for other codeine analogues.[31]
A 2014 report by the World Health Organizations Expert Committee on Drug Dependence found:
.. in many cases of tramadol dependence, a history of substance abuse is present.. but.. the evidence for physical dependence was considered minimal. Consequently, Tramadol is generally considered as a drug with low potential for dependence. In a recent German study (including a literature study, an analysis of two drug safety databases, and questionnaires analyses), the low abuse and low dependence potential of Tramadol were re-confirmed. The German expert group found a low prevalence of abuse or dependence in clinical practice in Germany, and concluded that Tramadol has a low potential for misuse, abuse, and dependence in Germany.[33]
Because of the possibility of convulsions at high doses for some users, recreational use can be very dangerous.[34] Tramadol can cause a higher incidence of nausea, dizziness, loss of appetite compared with opioids, which could deter recreational use.[35] Compared to hydrocodone, fewer persons choose to use tramadol recreationally.[36]
Overdose[edit]
Recognised risk factors for tramadol overdose include depression, addiction, and seizures.[37]Naloxone only partially reverses the toxic effects of tramadol overdose and may increase the risk of seizures.[15]
Deaths with tramadol overdose have been reported and are increasing in frequency in Northern Ireland; the majority of these overdoses involves other drugs including alcohol.[37] There were 254 tramadol-related deaths in England and Wales in 2013, and 379 in Florida in 2011.[38][39] In 2011, 21,649 emergency room visits in the United States were related to tramadol.[40]
Interactions[edit]
Tramadol may interact with certain antidepressants and anxiolytics (particularly selective serotonin reuptake inhibitors, serotonin–norepinephrine reuptake inhibitors, monoamine oxidase inhibitors, and tricyclic antidepressants) other opioid analgesics (pethidine, tapentadol, oxycodone, and fentanyl), dextromethorphan, certain migraine medications (triptans, ergots), certain antibiotics (namely, linezolid and isoniazid), certain herbs (e.g. St. John's wort, passiflora, etc.), stimulants (including amphetamines, phenethylamine, and phentermine), lithium, and methylene blue, as well as numerous other therapeutic agents.[7][15] As it is a substrate of CYP3A4 and CYP2D6, any agents with the ability to inhibit or induce these enzymes are likely interact with tramadol. A pressor response similar to the so-called 'cheese effect' was noted in combinations of amphetamine and tramadol, which appears to cause dysfunction of or toxicity to epinephrine/norepinephrine receptors.[15][25]Cyclobenzaprine, a commonly used muscle relaxant, atypical analgesic adjunct, as well as a potentiator often used with analgesics such as codeine, dihydrocodeine, hydrocodone and the like, is structurally related to the tricyclic antidepressants,[41] so should not be used with tramadol; this is also the case for trazodone.[42]
Pharmacology[edit]
Pharmacodynamics[edit]
Tramadol induces analgesic effects through a variety of different targets on the noradrenergic system, serotoninergic system and opioid receptors system.[43] Tramadol exists as a racemic mixture, the positive enantiomer inhibits serotonin reuptake while the negative enantiomer inhibits noradrenaline re-uptake, by binding to and blocking the transporters.[44][6] Tramadol has also been shown to act as a serotonin releasing agent. Both enantiomers of tramadol are agonists of the mu-opioid receptor and its M1 metabolite, O-demethylate, is also a mu-opoid receptor agonist but is 6 times more potent than tramadol itself.[45] All these effects work synergistically to induce analgesia.
Mechanism of action[edit]
| Site | Tramadol | DSMT | Species | Ref |
|---|---|---|---|---|
| MOR | 1,600–12,486 2,120–8,300 ≥1,000 (EC50) | 5.4–18.6 17 ((+)) ≥240 (EC50) | Human Rat Human | [49][50][51] [52][53] [54][11] |
| DOR | >10,000 57,600–100,000 | ≥2,900 690 (+)) | Human Rat | [49][50][55] [53][52] |
| KOR | >10,000 42,700–81,000 | ≥450 1,800 (+)) | Human Rat | [49][50][55] [53][52] |
| SERT | ~900 (IC50) 992–1,190 | >20,000 (IC50) 2,980 ((−)) (IC50) | Human Rat | [56] [53][11] |
| NET | 14,600 785 | 1,080 (−) (IC50) >860 (IC50) | Human Rat | [11] [53][11] |
| DAT | >100,000 | >20,000 | Rat | [57][55] |
| 5-HT1A | >20,000 | >20,000 | Rat | [55] |
| 5-HT2A | >20,000 | >20,000 | Rat | [55] |
| 5-HT2C | 1,000 (IC50) | 1,300 (IC50) | Rat | [58][59] |
| 5-HT3 | >20,000 | >20,000 | Rat | [55] |
| NK1 | IA | ? | Rat | [60][61] |
| M1 | >20,000 3,400 (IC50) | >20,000 2,000 (IC50) | Rat Multiple | [55] [62][63] |
| M2 | ND | ND | ND | ND |
| M3 | 1,000 (IC50) | IA | Human | [63][64] |
| M4 | ND | ND | ND | ND |
| M5 | ND | ND | ND | ND |
| α7 | 7,400 | ND | Chicken | [65] |
| σ1 | >10,000 | ND | Rat | [46][66] |
| σ2 | >10,000 | ND | Rat | [46] |
| NMDAR | 16,400 (IC50) | 16,500 (IC50) | Human | [67] |
| NMDAR (MK-801) | >20,000 | >20,000 | Rat | [55] |
| GABAA | >100,000 (IC50) | >100,000 (IC50) | Human | [67] |
| GlyR | >100,000 (IC50) | >100,000 (IC50) | Human | [67] |
| TRPA1 | 100– 10,000 (SI) | 1,000– 10,000 (SI) | Human | [68] |
| TRPV1 | >10,000 (IC50) | >10,000 (IC50) | Human | [68][69] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. | ||||
| Action | Value |
|---|---|
| 5-HT reuptake | 1,820 |
| 5-HT release | >10,000 |
| NE reuptake | 2,770 |
| NE release | >10,000 |
| DA reuptake | >10,000 |
| DA release | >10,000 |
| Values for reuptake inhibition are Ki (nM) and for release induction are EC50 (nM). | |
Tramadol has been found to possess these actions:[47][48][71]
- Agonist of the μ-opioid receptor (MOR) and to a far lesser extent of the δ-opioid receptor (DOR) and κ-opioid receptor (KOR)
- Serotonin reuptake inhibitor (SRI) and norepinephrine reuptake inhibitor); hence, an SNRI
- M1 and M3muscarinic acetylcholine receptor antagonist
- α7nicotinic acetylcholine receptor antagonist
- NMDA receptor antagonist (very weak)
Tramadol acts on the opioid receptors through its major active metabolitedesmetramadol, which has as much as 700-fold higher affinity for the MOR relative to tramadol.[11] Moreover, tramadol itself has been found to possess no efficacy in activating the MOR in functional activity assays, whereas desmetramadol activates the receptor with high intrinsic activity (Emax equal to that of morphine).[54][11][72] As such, desmetramadol is exclusively responsible for the opioid effects of tramadol.[73] Both tramadol and desmetramadol have pronounced selectivity for the MOR over the DOR and KOR in terms of binding affinity.[55][50][52]
Tramadol is well-established as an SRI.[47][48] In addition, a few studies have found that it also acts as a serotonin releasing agent (1–10 μM), similar in effect to fenfluramine.[74][75][76][77] The serotonin releasing effects of tramadol could be blocked by sufficiently high concentrations of the serotonin reuptake inhibitor 6-nitroquipazine, which is in accordance with other serotonin releasing agents such as fenfluramine and MDMA.[74][76][77] However, two more recent studies failed to find a releasing effect of tramadol at respective concentrations up to 10 and 30 μM.[78][77][70] In addition to serotonergic activity, tramadol is also a norepinephrine reuptake inhibitor.[47][48] It is not a norepinephrine releasing agent.[79][80][81][70] Tramadol does not inhibit the reuptake or induce the release of dopamine.[79][70]
A positron emission tomographyimaging study found that single oral 50-mg and 100-mg doses of tramadol to human volunteers resulted in 34.7% and 50.2% respective mean occupation of the serotonin transporter (SERT) in the thalamus.[82] The estimated median effective dose (ED50) for SERT occupancy hence was 98.1 mg, which was associated with a plasma tramadol level of about 330 ng/ml (1,300 nM).[82] The estimated maximum daily dosage of tramadol of 400 mg (100 mg q.i.d.) would result in as much as 78.7% occupancy of the SERT (in association with a plasma concentration of 1,220 ng/ml or 4,632 nM).[82] This is close to that of SSRIs, which occupy the SERT by 80% or more.[82]
Peak plasma concentrations during treatment with clinical dosages of tramadol have generally been found to be in the range of 70 to 592 ng/ml (266–2,250 nM) for tramadol and 55 to 143 ng/ml (221–573 nM) for desmetramadol.[83] The highest levels of tramadol were observed with the maximum oral daily dosage of 400 mg per day divided into one 100-mg dose every 6 hours (i.e., four 100-mg doses evenly spaced out per day).[83][84] Some accumulation of tramadol occurs with chronic administration; peak plasma levels with the maximum oral daily dosage (100 mg q.i.d.) are about 16% higher and the area-under-the-curve levels 36% higher than following a single oral 100-mg dose.[83]Positron emission tomography imaging studies have reportedly found that tramadol levels are at least four-fold higher in the brain than in plasma.[79][85] Conversely, brain levels of desmetramadol 'only slowly approach those in plasma'.[79] The plasma protein binding of tramadol is only 4 to 20%; hence, almost all tramadol in circulation is free, thus bioactive.[86][87][88]
Correspondence to effects[edit]
Co-administration of quinidine, a potent CYP2D6 enzyme inhibitor, with tramadol, a combination which results in markedly reduced levels of desmetramadol, was found not to significantly affect the analgesic effects of tramadol in human volunteers.[11][87] However, other studies have found that the analgesic effects of tramadol are significantly decreased or even absent in CYP2D6 poor metabolizers.[11][73] The analgesic effects of tramadol are only partially reversed by naloxone in human volunteers,[11] hence indicating that its opioid action is unlikely the sole factor; tramadol's analgesic effects are also partially reversed by α2-adrenergic receptor antagonists such as yohimbine, the 5-HT3 receptor antagonist ondansetron, and the 5-HT7 receptor antagonists SB-269970 and SB-258719.[16][89] Pharmacologically, tramadol is similar to tapentadol and methadone in that it not only binds to the MOR, but also inhibits the reuptake of serotonin and norepinephrine[4] due to its action on the noradrenergic and serotonergic systems, such as its 'atypical' opioid activity.[90]
Tramadol has inhibitory actions on the 5-HT2C receptor. Antagonism of 5-HT2C could be partially responsible for tramadol's reducing effect on depressive and obsessive–compulsive symptoms in patients with pain and co-morbid neurological illnesses.[58] 5-HT2C blockade may also account for its lowering of the seizure threshold, as 5-HT2Cknockout mice display significantly increased vulnerability to epileptic seizures, sometimes resulting in spontaneous death. However, the reduction of seizure threshold could be attributed to tramadol's putative inhibition of GABAA receptors at high doses (significant inhibition at 100 μM).[67][71] In addition, desmetramadol is a high-affinity ligand of the DOR, and activation of this receptor could be involved in tramadol's ability to provoke seizures in some individuals, as DOR agonists are well known for inducing seizures.[52]
Nausea and vomiting caused by tramadol are thought to be due to activation of the 5-HT3 receptor via increased serotonin levels.[56] In accordance, the 5-HT3 receptor antagonist metoclopramide can be used to treat tramadol-associated nausea and vomiting.[56] Tramadol and desmetramadol themselves do not bind to the 5-HT3 receptor.[56][48]
Pharmacokinetics[edit]
Tramadol undergoes hepatic metabolism via the cytochrome P450isozymeCYP2B6, CYP2D6, and CYP3A4, being O- and N-demethylated to five different metabolites. Of these, desmetramadol (O-desmethyltramadol) is the most significant, since it has 200 times the μ-affinity of (+)-tramadol, and furthermore has an elimination half-life of 9 hours, compared with 6 hours for tramadol itself. As with codeine, in the 6% of the population who have reduced CYP2D6 activity (hence reducing metabolism), a reduced analgesic effect is seen. Those with decreased CYP2D6 activity require a dose increase of 30% to achieve the same degree of pain relief as those with a normal level of CYP2D6 activity.[91][92]
Phase II hepatic metabolism renders the metabolites water-soluble, which are excreted by the kidneys. Thus, reduced doses may be used in renal and hepatic impairment.[16]
Its volume of distribution is around 306 l after oral administration and 203 l after parenteral administration.[16]
Chemistry[edit]
Tramadol is marketed as a racemic mixture of both R- and S-stereoisomers,[4] because the two isomers complement each other's analgesic activities.[4] The (+)-isomer is predominantly active as an opiate with a higher affinity for the µ-opiate receptor (20 times higher affinity than the (-)-isomer).[93]
Synthesis and stereoisomerism[edit]
| (1R,2R)-tramadol | (1S,2S)-tramadol |
| (1R,2S)-tramadol | (1S,2R)-tramadol |
The chemical synthesis of tramadol is described in the literature.[94] Tramadol [2-(dimethylaminomethyl)-1-(3-methoxyphenyl)cyclohexanol] has two stereogenic centers at the cyclohexane ring. Thus, 2-(dimethylaminomethyl)-1-(3-methoxyphenyl)cyclohexanol may exist in four different configurational forms:
- (1R,2R)-isomer
- (1S,2S)-isomer
- (1R,2S)-isomer
- (1S,2R)-isomer
The synthetic pathway leads to the racemate (1:1 mixture) of (1R,2R)-isomer and the (1S,2S)-isomer as the main products. Minor amounts of the racemic mixture of the (1R,2S)-isomer and the (1S,2R)-isomer are formed as well. The isolation of the (1R,2R)-isomer and the (1S,2S)-isomer from the diastereomeric minor racemate [(1R,2S)-isomer and (1S,2R)-isomer] is realized by the recrystallization of the hydrochlorides.The drug tramadol is a racemate of the hydrochlorides of the (1R,2R)-(+)- and the (1S,2S)-(−)-enantiomers.The resolution of the racemate [(1R,2R)-(+)-isomer / (1S,2S)-(−)-isomer] was described[95] employing (R)-(−)- or (S)-(+)-mandelic acid. This process does not find industrial application, since tramadol is used as a racemate, despite known different physiological effects[96] of the (1R,2R)- and (1S,2S)-isomers, because the racemate showed higher analgesic activity than either enantiomer in animals[97] and in humans.[98]
Detection in biological fluids[edit]
Tramadol and desmetramadol may be quantified in blood, plasma or serum to monitor for abuse, confirm a diagnosis of poisoning or assist in the forensic investigation of a sudden death. Most commercial opiate immunoassay screening tests do not cross-react significantly with tramadol or its major metabolites, so chromatographic techniques must be used to detect and quantitate these substances. The concentration of desmetramadol in the blood or plasma of a person who has taken tramadol is generally 10–20% those of the parent drug.[99][100][101]
Society and culture[edit]
Patent history[edit]
The U.S. Food and Drug Administration (FDA) approved tramadol in March 1995 and an extended-release (ER) formulation in September 2005.[102] ER Tramadol was protected by US patents nos. 6,254,887[103] and 7,074,430.[104][105] The FDA listed the patents' expiration as 10 May 2014.[104] However, in August 2009, US District Court for the District of Delaware ruled the patents invalid, a decision upheld the following year by the Court of Appeals for the Federal Circuit. Manufacture and distribution of generic equivalents of Ultram ER in the United States was therefore permitted prior to the expiration of the patents.[106]
Legal status[edit]
Effective August 18, 2014, tramadol has been placed into Schedule IV of the federal Controlled Substances Act in the United States.[107][108] Before that, some US states had already classified tramadol as a Schedule IV controlled substance under their respective state laws.[109][110][111]
Tramadol is classified in Schedule 4 (prescription only) in Australia, rather than as a Schedule 8 Controlled Drug (Possession without authority illegal) like most other opioids.[15]
Effective May 2008, Sweden classified tramadol as a controlled substance in the same category as codeine and dextropropoxyphene, but allows a normal prescription be used currently.[112]
The UK classified tramadol as a Class C, Schedule 3 controlled drug on 10 June 2014, but exempted it from the safe custody requirement.[113]
Misuse[edit]
Illicit use of the drug is thought to be a major factor in the success of the Boko Haram terrorist organization.[114][115][116] When used at higher doses, the drug 'can produce similar effects to heroin.'[114] Said one former member, “whenever we took tramadol, nothing mattered to us anymore except what we were sent to do because it made us very high and very bold, it was impossible to go on a mission without taking it.”[114] Tramadol misuse is also found as a coping mechanism in the Gaza Strip.[117]
Research[edit]
Investigational uses[edit]
- Diabetic neuropathy[118][119]
- Antidepressant[120]
- Postherpetic neuralgia[121][122]
- Premature ejaculation[123][124]
- Adjunct to local anaesthesia[125]
False findings about sources in nature[edit]
In 2013, researchers reported that tramadol was found in relatively high concentrations (1%+) in the roots of the African pin cushion tree (Nauclea latifolia).[126] In 2014, however, it was reported that the presence of tramadol in the tree roots was the result of tramadol having been administered to cattle by farmers in the region:[127] tramadol and its metabolites were present in the animals' excreta, which contaminated the soil around the trees. Therefore, tramadol and its mammalian metabolites were found in tree roots in the far north of Cameroon, but not in the south where it is not administered to farm animals.[127]
A 2014 editorial in Lab Times online contested the notion that tramadol in tree roots was the result of anthropogenic contamination, stating that samples were taken from trees which grew in national parks, where livestock were forbidden; it also quoted researcher Michel de Waard, who stated that 'thousands and thousands of tramadol-treated cattle sitting around a single tree and urinating there' would be required to produce the concentrations discovered.[128]
In 2015, radiocarbon analysis confirmed that the tramadol found in N.latifolia roots could not be plant-derived and was of synthetic origin.[129]
Veterinary medicine[edit]
Tramadol may be used to treat post-operative, injury-related, and chronic (e.g., cancer-related) pain in dogs and cats as well as rabbits, coatis, many small mammals including rats and flying squirrels, guinea pigs, ferrets, and raccoons.[130]
| Species | Half-life (h) for parent drug | Half-life (h) for desmetramadol | Maximum plasma concentration (ng/mL) for parent drug | Maximum plasma concentration (ng/mL) for desmetramadol |
|---|---|---|---|---|
| Camel | 3.2 (IM), 1.3 (IV) | – | 0.44 (IV) | – |
| Cat | 3.40 (oral), 2.23 (IV) | 4.82 (oral), 4.35 (IV) | 914 (oral), 1323 (IV) | 655 (oral), 366 (IV) |
| Dog | 1.71 (oral), 1.80 (IV), 2.24 (rectal) | 2.18 (oral), 90-5000 (IV) | 1402.75 (oral) | 449.13 (oral), 90–350 (IV) |
| Donkey | 4.2 (oral), 1.5 (IV) | – | 2817 (oral) | – |
| Goat | 2.67 (oral), 0.94 (IV) | – | 542.9 (oral) | – |
| Horses | 1.29–1.53 (IV), 10.1 (oral) | 4 (oral) | 637 (IV), 256 (oral) | 47 (oral) |
| Llama | 2.54 (IM), 2.12 (IV) | 7.73 (IM), 10.4 (IV) | 4036 (IV), 1360 (IM) | 158 (IV), 158 (IM) |
References[edit]
- ^ abcdefghijklmn'Tramadol Hydrochloride'. The American Society of Health-System Pharmacists. Retrieved 1 December 2014.
- ^ abc'Tramadol'. Drugs.com. Retrieved 22 December 2018.
- ^'Tramadol: MedlinePlus Drug Information'. medlineplus.gov.
- ^ abcdeBrayfield, A, ed. (13 December 2013). 'Tramadol Hydrochloride'. Martindale: The Complete Drug Reference. Pharmaceutical Press. Retrieved 5 April 2014.
- ^ ab'Ultram, Ultram ER (tramadol) dosing, indications, interactions, adverse effects, and more'. Medscape Reference. WebMD. Retrieved 28 November 2013.
- ^ abDayer, P; Desmeules, J; Collart, L (1997). '[Pharmacology of tramadol]'. Drugs. 53 Suppl 2: 18–24. doi:10.2165/00003495-199700532-00006. PMID9190321.
- ^ abc'Australian Label: Tramadol Sandoz 50 mg capsules'(PDF). TGA eBusiness Services. 4 November 2011. Retrieved 6 April 2014.
- ^ abcBritish national formulary : BNF 74 (74 ed.). British Medical Association. 2017. pp. 447–448. ISBN978-0857112989.
- ^'Tramadol Pregnancy and Breastfeeding Warnings'. Drugs.com. Retrieved 5 September 2016.
- ^ abcdLeppert W (November – December 2009). 'Tramadol as an analgesic for mild to moderate cancer pain'(PDF). Pharmacological Reports. 61 (6): 978–92. doi:10.1016/s1734-1140(09)70159-8. PMID20081232.
- ^ abcdefghijRaffa RB, Buschmann H, Christoph T, Eichenbaum G, Englberger W, Flores CM, Hertrampf T, Kögel B, Schiene K, Straßburger W, Terlinden R, Tzschentke TM (2012). 'Mechanistic and functional differentiation of tapentadol and tramadol'. Expert Opin Pharmacother. 13 (10): 1437–49. doi:10.1517/14656566.2012.696097. PMID22698264.
- ^Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 528. ISBN9783527607495.
- ^'NADAC as of 2018-12-19'. Centers for Medicare and Medicaid Services. Retrieved 22 December 2018.
- ^'The Top 300 of 2019'. clincalc.com. Retrieved 22 December 2018.
- ^ abcdefghijklRossi, S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN978-0-9805790-9-3.
- ^ abcdefGrond S, Sablotzki A (2004). 'Clinical pharmacology of tramadol'. Clinical Pharmacokinetics. 43 (13): 879–923. doi:10.2165/00003088-200443130-00004. PMID15509185.
- ^Lee CR, McTavish D, Sorkin EM (1993). 'Tramadol. A preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in acute and chronic pain states'. Drugs. 46 (2): 313–40. doi:10.2165/00003495-199346020-00008. PMID7691519.
- ^ abMacLean AJ, Schwartz TL (May 2015). 'Tramadol for the treatment of fibromyalgia'. Expert Rev Neurother. 15 (5): 469–75. doi:10.1586/14737175.2015.1034693. PMID25896486.
- ^ abcdeBloor M, Paech MJ, Kaye R (2012). 'Tramadol in pregnancy and lactation'. International Journal of Obstetric Anesthesia. 21 (2): 163–67. doi:10.1016/j.ijoa.2011.10.008. PMID22317891.
- ^'FDA Drug Safety Communication: FDA evaluating the risks of using the pain medicine tramadol in children aged 17 and younger'. FDA. FDA Drug Safety and Availability. Retrieved 21 September 2015.
- ^Commissioner, Office of the. 'Press Announcements - FDA statement from Douglas Throckmorton, M.D., deputy center director for regulatory programs, Center for Drug Evaluation and Research, on new warnings about the use of codeine and tramadol in children & nursing mothers'. www.fda.gov. Retrieved 21 April 2017.
- ^'FDA Drug Safety Communication: FDA restricts use of prescription codeine pain and cough medicines and tramadol pain medicines in children; recommends against use in breastfeeding women'. Food and Drug Administration.
- ^'Tramadol'. MedlinePlus. American Society of Health-System Pharmacists. 1 September 2008. Retrieved 29 September 2009.
- ^Langley PC, Patkar AD, Boswell KA, Benson CJ, Schein JR (2010). 'Adverse event profile of tramadol in recent clinical studies of chronic osteoarthritis pain'. Current Medical Research and Opinion. 26 (1): 239–51. doi:10.1185/03007990903426787. PMID19929615.
- ^ abcKeating GM (2006). 'Tramadol sustained-release capsules'. Drugs. 66 (2): 223–30. doi:10.2165/00003495-200666020-00006. PMID16451094.
- ^Bryant et al. 1988 and Rouveix 1992 cited by Collett BJ (July 2001). 'Chronic opioid therapy for non-cancer pain'. British Journal of Anaesthesia. 87 (1): 133–43. doi:10.1093/bja/87.1.133. PMID11460802.
- ^Sacerdote P, Bianchi M, Gaspani L, Manfredi B, Maucione A, Terno G, Ammatuna M, Panerai AE (2000). 'The effects of tramadol and morphine on immune responses and pain after surgery in cancer patients'(PDF). Anesthesia & Analgesia. 90 (6): 1411–14. doi:10.1097/00000539-200006000-00028. PMID10825330.
- ^Liu Z, Gao F, Tian Y (2006). 'Effects of morphine, fentanyl and tramadol on human immune response'. J. Huazhong Univ. Sci. Technol. Med. Sci. 26 (4): 478–81. doi:10.1007/s11596-006-0427-5. PMID17120754.
- ^Sacerdote P, Bianchi M, Manfredi B, Panerai AE (1997). 'Effects of tramadol on immune responses and nociceptive thresholds in mice'. Pain. 72 (3): 325–30. doi:10.1016/S0304-3959(97)00055-9. PMID9313273.
- ^'Withdrawal syndrome and dependence: tramadol too'. Prescrire Int. 12 (65): 99–100. 2003. PMID12825576.
- ^ abcEpstein DH, Preston KL, Jasinski DR (2006). 'Abuse liability, behavioral pharmacology, and physical-dependence potential of opioids in humans and laboratory animals: lessons from tramadol'. Biological Psychology. 73 (1): 90–99. doi:10.1016/j.biopsycho.2006.01.010. PMC2943845. PMID16497429.
- ^Senay EC, Adams EH, Geller A, Inciardi JA, Muñoz A, Schnoll SH, Woody GE, Cicero TJ (April 2003). 'Physical dependence on Ultram (tramadol hydrochloride): both opioid-like and atypical withdrawal symptoms occur'. Drug Alcohol Depend. 69 (3): 233–41. CiteSeerX10.1.1.524.5426. doi:10.1016/S0376-8716(02)00321-6. PMID12633909.
- ^Tramadol - Update Review Report. WHO (16‐20 June 2014)
- ^Jovanović-Cupić V, Martinović Z, Nesić N (2006). 'Seizures associated with intoxication and abuse of tramadol'. Clinical Toxicology. 44 (2): 143–46. doi:10.1080/1556365050014418. PMID16615669.
- ^Rodriguez RF, Bravo LE, Castro F, Montoya O, Castillo JM, Castillo MP, Daza P, Restrepo JM, Rodriguez MF (2007). 'Incidence of weak opioids adverse events in the management of cancer pain: a double-blind comparative trial'. Journal of Palliative Medicine. 10 (1): 56–60. doi:10.1089/jpm.2006.0117. PMID17298254.
- ^Adams EH, Breiner S, Cicero TJ, Geller A, Inciardi JA, Schnoll SH, Senay EC, Woody GE (2006). 'A comparison of the abuse liability of tramadol, NSAIDs, and hydrocodone in patients with chronic pain'. Journal of Pain and Symptom Management. 31 (5): 465–76. doi:10.1016/j.jpainsymman.2005.10.006. PMID16716877.
- ^ abRandall C, Crane J (2014). 'Tramadol deaths in Northern Ireland: a review of cases from 1996 to 2012'. Journal of Forensic and Legal Medicine. 23: 32–36. doi:10.1016/j.jflm.2014.01.006. PMID24661703.
- ^White M. 'Tramadol Deaths in the United Kingdom'(pdf_e). Public Health England.
- ^Fauber J (22 December 2013). 'Killing Pain: Tramadol the 'Safe' Drug of Abuse'.
- ^Scheck J (19 October 2016). 'Tramadol: The Opioid Crisis for the rest of the World'. The Wall Street Journal. Dow Jones & Co. Retrieved 4 January 2019.
- ^'cyclobenzaprine oral : Uses, Side Effects, Interactions, Pictures, Warnings & Dosing'. WebMD. Retrieved 8 November 2016.
- ^'Trazodone: Common sleep drug is little-known antidepressant'. Consumer Reports. Retrieved 8 November 2016.
- ^Hitchings, Andrew; Lonsdale, Dagan; Burrage, Daniel; Baker, Emma (2015). Top 100 drugs : clinical pharmacology and practical prescribing. Churchill Livingstone Elsevier. pp. 168–169. ISBN978-0-7020-5516-4.
- ^Vazzana, M; Andreani, T; Fangueiro, J; Faggio, C; Silva, C; Santini, A; Garcia, ML; Silva, AM; Souto, EB (March 2015). 'Tramadol hydrochloride: pharmacokinetics, pharmacodynamics, adverse side effects, co-administration of drugs and new drug delivery systems'. Biomedicine & Pharmacotherapy. 70: 234–8. doi:10.1016/j.biopha.2015.01.022. PMID25776506.
- ^'Tramadol'. www.drugbank.ca.
- ^ abcRoth, BL; Driscol, J. 'PDSP Ki Database'. Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 14 August 2017.
- ^ abcdMinami K, Uezono Y, Ueta Y (2007). 'Pharmacological aspects of the effects of tramadol on G-protein coupled receptors'. J. Pharmacol. Sci. 103 (3): 253–60. doi:10.1254/jphs.cr0060032. PMID17380034.
- ^ abcdeMinami K, Ogata J, Uezono Y (2015). 'What is the main mechanism of tramadol?'. Naunyn Schmiedebergs Arch. Pharmacol. 388 (10): 999–1007. doi:10.1007/s00210-015-1167-5. PMID26292636.
- ^ abcWentland MP, Lou R, Lu Q, Bu Y, VanAlstine MA, Cohen DJ, Bidlack JM (2009). 'Syntheses and opioid receptor binding properties of carboxamido-substituted opioids'. Bioorg. Med. Chem. Lett. 19 (1): 203–8. doi:10.1016/j.bmcl.2008.10.134. PMID19027293.
- ^ abcdShen Q, Qian Y, Huang X, Xu X, Li W, Liu J, Fu W (2016). 'Discovery of Potent and Selective Agonists of δ Opioid Receptor by Revisiting the 'Message-Address' Concept'. ACS Med Chem Lett. 7 (4): 391–6. doi:10.1021/acsmedchemlett.5b00423. PMC4834657. PMID27096047.
- ^Volpe DA, McMahon Tobin GA, Mellon RD, Katki AG, Parker RJ, Colatsky T, Kropp TJ, Verbois SL (2011). 'Uniform assessment and ranking of opioid μ receptor binding constants for selected opioid drugs'. Regul. Toxicol. Pharmacol. 59 (3): 385–90. doi:10.1016/j.yrtph.2010.12.007. PMID21215785.
- ^ abcdePotschka H, Friderichs E, Löscher W (September 2000). 'Anticonvulsant and proconvulsant effects of tramadol, its enantiomers and its M1 metabolite in the rat kindling model of epilepsy'. Br. J. Pharmacol. 131 (2): 203–12. doi:10.1038/sj.bjp.0703562. PMC1572317. PMID10991912.
- ^ abcdeCodd EE, Shank RP, Schupsky JJ, Raffa RB (1995). 'Serotonin and norepinephrine uptake inhibiting activity of centrally acting analgesics: structural determinants and role in antinociception'. J. Pharmacol. Exp. Ther. 274 (3): 1263–70. PMID7562497.
- ^ abGillen C, Haurand M, Kobelt DJ, Wnendt S (2000). 'Affinity, potency and efficacy of tramadol and its metabolites at the cloned human mu-opioid receptor'. Naunyn Schmiedebergs Arch. Pharmacol. 362 (2): 116–21. doi:10.1007/s002100000266. PMID10961373.
- ^ abcdefghiFrink MC, Hennies HH, Englberger W, Haurand M, Wilffert B (1996). 'Influence of tramadol on neurotransmitter systems of the rat brain'. Arzneimittelforschung. 46 (11): 1029–36. PMID8955860.
- ^ abcdBarann M, Urban B, Stamer U, Dorner Z, Bönisch H, Brüss M (2006). 'Effects of tramadol and O-demethyl-tramadol on human 5-HT reuptake carriers and human 5-HT3A receptors: a possible mechanism for tramadol-induced early emesis'. Eur. J. Pharmacol. 531 (1–3): 54–8. doi:10.1016/j.ejphar.2005.11.054. PMID16427041.
- ^Raffa RB, Friderichs E, Reimann W, Shank RP, Codd EE, Vaught JL (1992). 'Opioid and nonopioid components independently contribute to the mechanism of action of tramadol, an 'atypical' opioid analgesic'. J. Pharmacol. Exp. Ther. 260 (1): 275–85. PMID1309873.
- ^ abOgata J, Minami K, Uezono Y, Okamoto T, Shiraishi M, Shigematsu A, Ueta Y (2004). 'The inhibitory effects of tramadol on 5-hydroxytryptamine type 2C receptors expressed in Xenopus oocytes'. Anesth. Analg. 98 (5): 1401–6, table of contents. doi:10.1213/01.ANE.0000108963.77623.A4. PMID15105221.
- ^Horishita T, Minami K, Uezono Y, Shiraishi M, Ogata J, Okamoto T, Shigematsu A (2006). 'The tramadol metabolite, O-desmethyl tramadol, inhibits 5-hydroxytryptamine type 2C receptors expressed in Xenopus Oocytes'. Pharmacology. 77 (2): 93–9. doi:10.1159/000093179. PMID16679816.
- ^Okamoto T, Minami K, Uezono Y, Ogata J, Shiraishi M, Shigematsu A, Ueta Y (2003). 'The inhibitory effects of ketamine and pentobarbital on substance p receptors expressed in Xenopus oocytes'. Anesth. Analg. 97 (1): 104–10, table of contents. doi:10.1213/01.ANE.0000066260.99680.11. PMID12818951.
- ^Minami K, Yokoyama T, Ogata J, Uezono Y (2011). 'The tramadol metabolite O-desmethyl tramadol inhibits substance P-receptor functions expressed in Xenopus oocytes'. J. Pharmacol. Sci. 115 (3): 421–4. doi:10.1254/jphs.10313sc. PMID21372504.
- ^Shiraishi M, Minami K, Uezono Y, Yanagihara N, Shigematsu A (2001). 'Inhibition by tramadol of muscarinic receptor-induced responses in cultured adrenal medullary cells and in Xenopus laevis oocytes expressing cloned M1 receptors'. J. Pharmacol. Exp. Ther. 299 (1): 255–60. PMID11561087.
- ^ abNakamura M, Minami K, Uezono Y, Horishita T, Ogata J, Shiraishi M, Okamoto T, Terada T, Sata T (2005). 'The effects of the tramadol metabolite O-desmethyl tramadol on muscarinic receptor-induced responses in Xenopus oocytes expressing cloned M1 or M3 receptors'. Anesth. Analg. 101 (1): 180–6, table of contents. doi:10.1213/01.ANE.0000154303.93909.A3. PMID15976229.
- ^Shiga Y, Minami K, Shiraishi M, Uezono Y, Murasaki O, Kaibara M, Shigematsu A (2002). 'The inhibitory effects of tramadol on muscarinic receptor-induced responses in Xenopus oocytes expressing cloned M(3) receptors'. Anesth. Analg. 95 (5): 1269–73, table of contents. doi:10.1097/00000539-200211000-00031. PMID12401609.
- ^Shiraishi M, Minami K, Uezono Y, Yanagihara N, Shigematsu A, Shibuya I (2002). 'Inhibitory effects of tramadol on nicotinic acetylcholine receptors in adrenal chromaffin cells and in Xenopus oocytes expressing alpha 7 receptors'. Br. J. Pharmacol. 136 (2): 207–16. doi:10.1038/sj.bjp.0704703. PMC1573343. PMID12010769.
- ^Sánchez-Fernández C, Montilla-García Á, González-Cano R, Nieto FR, Romero L, Artacho-Cordón A, Montes R, Fernández-Pastor B, Merlos M, Baeyens JM, Entrena JM, Cobos EJ (2014). 'Modulation of peripheral μ-opioid analgesia by σ1 receptors'. J. Pharmacol. Exp. Ther. 348 (1): 32–45. doi:10.1124/jpet.113.208272. PMID24155346.
- ^ abcdHara K, Minami K, Sata T (2005). 'The effects of tramadol and its metabolite on glycine, gamma-aminobutyric acidA, and N-methyl-D-aspartate receptors expressed in Xenopus oocytes'. Anesth. Analg. 100 (5): 1400–5, table of contents. doi:10.1213/01.ANE.0000150961.24747.98. PMID15845694.
- ^ abMiyano K, Minami K, Yokoyama T, Ohbuchi K, Yamaguchi T, Murakami S, Shiraishi S, Yamamoto M, Matoba M, Uezono Y (2015). 'Tramadol and its metabolite m1 selectively suppress transient receptor potential ankyrin 1 activity, but not transient receptor potential vanilloid 1 activity'. Anesth. Analg. 120 (4): 790–8. doi:10.1213/ANE.0000000000000625. PMID25642661.
- ^Marincsák R, Tóth BI, Czifra G, Szabó T, Kovács L, Bíró T (2008). 'The analgesic drug, tramadol, acts as an agonist of the transient receptor potential vanilloid-1'. Anesth. Analg. 106 (6): 1890–6. doi:10.1213/ane.0b013e318172fefc. PMID18499628.
- ^ abcdRothman RB, Baumann MH (2006). 'Therapeutic potential of monoamine transporter substrates'. Curr Top Med Chem. 6 (17): 1845–59. doi:10.2174/156802606778249766. PMID17017961.
- ^ abVazzana M, Andreani T, Fangueiro J, Faggio C, Silva C, Santini A, Garcia ML, Silva AM, Souto EB (2015). 'Tramadol hydrochloride: pharmacokinetics, pharmacodynamics, adverse side effects, co-administration of drugs and new drug delivery systems'. Biomed. Pharmacother. 70: 234–8. doi:10.1016/j.biopha.2015.01.022. PMID25776506.
- ^Minami K, Sudo Y, Miyano K, Murphy RS, Uezono Y (2015). 'µ-Opioid receptor activation by tramadol and O-desmethyltramadol (M1)'. J Anesth. 29 (3): 475–479. doi:10.1007/s00540-014-1946-z. PMID25394761.
- ^ abColler JK, Christrup LL, Somogyi AA (2009). 'Role of active metabolites in the use of opioids'. Eur. J. Clin. Pharmacol. 65 (2): 121–39. doi:10.1007/s00228-008-0570-y. PMID18958460.
- ^ abDriessen B, Reimann W (January 1992). 'Interaction of the central analgesic, tramadol, with the uptake and release of 5-hydroxytryptamine in the rat brain in vitro'. British Journal of Pharmacology. 105 (1): 147–51. doi:10.1111/j.1476-5381.1992.tb14226.x. PMC1908625. PMID1596676.
- ^Bamigbade TA, Davidson C, Langford RM, Stamford JA (September 1997). 'Actions of tramadol, its enantiomers and principal metabolite, O-desmethyltramadol, on serotonin (5-HT) efflux and uptake in the rat dorsal raphe nucleus'. British Journal of Anaesthesia. 79 (3): 352–56. doi:10.1093/bja/79.3.352. PMID9389855.
- ^ abReimann W, Schneider F (May 1998). 'Induction of 5-hydroxytryptamine release by tramadol, fenfluramine and reserpine'. European Journal of Pharmacology. 349 (2–3): 199–203. doi:10.1016/S0014-2999(98)00195-2. PMID9671098.
- ^ abcGobbi M, Moia M, Pirona L, Ceglia I, Reyes-Parada M, Scorza C, Mennini T (September 2002). 'p-Methylthioamphetamine and 1-(m-chlorophenyl)piperazine, two non-neurotoxic 5-HT releasers in vivo, differ from neurotoxic amphetamine derivatives in their mode of action at 5-HT nerve endings in vitro'. Journal of Neurochemistry. 82 (6): 1435–43. doi:10.1046/j.1471-4159.2002.01073.x. PMID12354291.
- ^Gobbi M, Mennini T (1999). 'Release studies with rat brain cortical synaptosomes indicate that tramadol is a 5-hydroxytryptamine uptake blocker and not a 5-hydroxytryptamine releaser'. Eur. J. Pharmacol. 370 (1): 23–6. doi:10.1016/s0014-2999(99)00123-5. PMID10323276.
- ^ abcdDriessen B, Reimann W, Giertz H (1993). 'Effects of the central analgesic tramadol on the uptake and release of noradrenaline and dopamine in vitro'. Br. J. Pharmacol. 108 (3): 806–11. doi:10.1111/j.1476-5381.1993.tb12882.x. PMC1908052. PMID8467366.
- ^Reimann W, Hennies HH (1994). 'Inhibition of spinal noradrenaline uptake in rats by the centrally acting analgesic tramadol'. Biochem. Pharmacol. 47 (12): 2289–93. doi:10.1016/0006-2952(94)90267-4. PMID8031323.
- ^Halfpenny DM, Callado LF, Hopwood SE, Bamigbade TA, Langford RM, Stamford JA (1999). 'Effects of tramadol stereoisomers on norepinephrine efflux and uptake in the rat locus coeruleus measured by real time voltammetry'. Br J Anaesth. 83 (6): 909–15. doi:10.1093/bja/83.6.909. PMID10700792.
- ^ abcdOgawa K, Tateno A, Arakawa R, Sakayori T, Ikeda Y, Suzuki H, Okubo Y (2014). 'Occupancy of serotonin transporter by tramadol: a positron emission tomography study with [11C]DASB'. Int. J. Neuropsychopharmacol. 17 (6): 845–50. doi:10.1017/S1461145713001764. PMID24423243.
- ^ abcGrond S, Sablotzki A (2004). 'Clinical pharmacology of tramadol'. Clin Pharmacokinet. 43 (13): 879–923. doi:10.2165/00003088-200443130-00004. PMID15509185.
- ^'Tramadol Dosage Guide with Precautions'.
- ^Tao Q, Stone DJ, Borenstein MR, Codd EE, Coogan TP, Desai-Krieger D, Liao S, Raffa RB (2002). 'Differential tramadol and O-desmethyl metabolite levels in brain vs. plasma of mice and rats administered tramadol hydrochloride orally'. J Clin Pharm Ther. 27 (2): 99–106. doi:10.1046/j.1365-2710.2002.00384.x. PMID11975693.
- ^Gibson TP (1996). 'Pharmacokinetics, efficacy, and safety of analgesia with a focus on tramadol HCl'. Am. J. Med. 101 (1A): 47S–53S. doi:10.1016/s0002-9343(96)90035-2. PMID8764760.
- ^ abDayer P, Collart L, Desmeules J (1994). 'The pharmacology of tramadol'. Drugs. 47 Suppl 1: 3–7. doi:10.2165/00003495-199400471-00003. PMID7517823.
- ^Nobilis M, Kopecký J, Kvetina J, Chládek J, Svoboda Z, Vorísek V, Perlík F, Pour M, Kunes J (March 2002). 'High-performance liquid chromatographic determination of tramadol and its O-desmethylated metabolite in blood plasma. Application to a bioequivalence study in humans'. J Chromatogr A. 949 (1–2): 11–22. doi:10.1016/S0021-9673(01)01567-9. PMID11999728.
- ^Yanarates O, Dogrul A, Yildirim V, Sahin A, Sizlan A, Seyrek M, Akgül O, Kozak O, Kurt E, Aypar U (2010). 'Spinal 5-HT7 receptors play an important role in the antinociceptive and antihyperalgesic effects of tramadol and its metabolite, O-Desmethyltramadol, via activation of descending serotonergic pathways'(PDF). Anesthesiology. 112 (3): 696–710. doi:10.1097/ALN.0b013e3181cd7920. PMID20179508.
- ^Micó JA, Ardid D, Berrocoso E, Eschalier A (2006). 'Antidepressants and pain'. Trends in Pharmacological Sciences. 27 (7): 348–54. doi:10.1016/j.tips.2006.05.004. PMID16762426.
- ^Leppert W (2011). 'CYP2D6 in the metabolism of opioids for mild to moderate pain'. Pharmacology. 87 (5–6): 274–85. doi:10.1159/000326085. PMID21494059.
- ^Samer CF, Lorenzini KI, Rollason V, Daali Y, Desmeules JA (2013). 'Applications of CYP450 testing in the clinical setting'. Molecular Diagnosis & Therapy. 17 (3): 165–84. doi:10.1007/s40291-013-0028-5. PMC3663206. PMID23588782.
- ^'Tramadol Hydrochloride 50mg Capsules'. UK Electronic Medicines Compendium. January 2016. Retrieved 16 March 2017.
- ^Pharmaceutical Substances, Axel Kleemann, Jürgen Engel, Bernd Kutscher and Dieter Reichert, 4. ed. (2000) 2 volumes, Thieme-Verlag Stuttgart (Germany), p. 2085 bis 2086, ISBN978-1-58890-031-9; since 2003 online with biannual actualizations.
- ^Zynovy Z, Meckler H (2000). 'A Practical Procedure for the Resolution of (+)- and (−)-Tramadol'. Organic Process Research & Development. 4 (4): 291–294. doi:10.1021/op000281v.
- ^Burke D, Henderson DJ (2002). 'Chirality: a blueprint for the future'. British Journal of Anaesthesia. 88 (4): 563–76. doi:10.1093/bja/88.4.563. PMID12066734.
- ^Raffa RB, Friderichs E, Reimann W, Shank RP, Codd EE, Vaught JL, Jacoby HI, Selve N (1993). 'Complementary and synergistic antinociceptive interaction between the enantiomers of tramadol'. J. Pharmacol. Exp. Ther. 267 (1): 331–40. PMID8229760.
- ^Grond S, Meuser T, Zech D, Hennig U, Lehmann KA (1995). 'Analgesic efficacy and safety of tramadol enantiomers in comparison with the racemate: a randomised, double-blind study with gynaecological patients using intravenous patient-controlled analgesia'. Pain. 62 (3): 313–20. doi:10.1016/0304-3959(94)00274-I. PMID8657431.
- ^Karhu D, El-Jammal A, Dupain T, Gaulin D, Bouchard S (2007). 'Pharmacokinetics and dose proportionality of three Tramadol Contramid OAD tablet strengths'. Biopharmaceutics & Drug Disposition. 28 (6): 323–30. doi:10.1002/bdd.561. PMID17575561.
- ^Tjäderborn M, Jönsson AK, Hägg S, Ahlner J (2007). 'Fatal unintentional intoxications with tramadol during 1995–2005'. Forensic Sci. Int. 173 (2–3): 107–11. doi:10.1016/j.forsciint.2007.02.007. PMID17350197.
- ^Baselt, R. (2011) Disposition of Toxic Drugs and Chemicals in Man, 9th edition, Biomedical Publications, Seal Beach, CA, pp. 1712–15, ISBN978-0-9626523-8-7.
- ^McCarberg B (2007). 'Tramadol extended-release in the management of chronic pain'. Therapeutics and Clinical Risk Management. 3 (3): 401–10. PMC2386353. PMID18488071.
- ^US patent 6254887, Miller RB, Leslie ST, Malkowska ST, Smith KJ, Wimmer S, Winkler H, Hahn U, Prater DA, 'Controlled Release Tramadol', issued 3 July 2001
- ^ abFDA AccessData entry for Tramadol Hydrochloride. Retrieved 17 August 2009.
- ^US patent 7074430, Miller RB, Malkowska ST, Wimmer S, Hahn U, Leslie ST, Smith KJ, Winkler H, Prater DA, 'Controlled Release Tramadol Tramadol Formulation', issued 11 July 2006
- ^Purdue Pharma Prods. L.P. v. Par Pharm., Inc., 377 Fed.Appx. 978 (Fed. Cir. 2010).
- ^'DEA controls tramadol as a schedule IV controlled substance effective August 18, 2014'. FDA Law Blog. 2 July 2014.
- ^'Federal Registrar'(PDF). gpo.gov.
- ^'TRAMADOL (Trade Names: Ultram®, Ultracet®)'. Drug Enforcement Administration (February 2011)
- ^'Tennessee News: Tramadol and Carisoprodol Now Classified Schedule IV'. National Association of Boards of Pharmacy (8 June 2011). Retrieved on 2012-12-26.
- ^'State of Ohio Board of Pharmacy'(PDF). Pharmacy.ohio.gov. 18 August 2014. Archived from the original(PDF) on 29 December 2016. Retrieved 8 November 2016.
- ^'Substansen tramadol nu narkotikaklassad på samma sätt som kodein och dextropropoxifen'. Lakemedelsverket. 14 May 2008. Retrieved 18 April 2010.[permanent dead link]
- ^'Tramadol to become a Controlled Drug in the UK'. vetdispense.co.uk. 2 June 2014. Retrieved 3 June 2014.
- ^ abc''If you take Tramadol away, you make Boko Haram weak.''. African Arguments. 15 March 2019. Retrieved 18 March 2019.
- ^'Drugs for war: Opioid abuse in West Africa'. BBC News. Retrieved 18 March 2019.
- ^www.csis.orghttps://www.csis.org/npfp/dangerous-opioid-india. Retrieved 18 March 2019.Missing or empty
|title=(help) - ^Berger, Miriam (7 January 2019). 'Gaza's Opioid Problem'. ISSN0027-8378. Retrieved 18 March 2019.
- ^Harati Y, Gooch C, Swenson M, Edelman S, Greene D, Raskin P, Donofrio P, Cornblath D, Sachdeo R, Siu CO, Kamin M (1998). 'Double-blind randomized trial of tramadol for the treatment of the pain of diabetic neuropathy'. Neurology. 50 (6): 1842–46. doi:10.1212/WNL.50.6.1842. PMID9633738.
- ^Harati Y, Gooch C, Swenson M, Edelman SV, Greene D, Raskin P, Donofrio P, Cornblath D, Olson WH, Kamin M (2000). 'Maintenance of the long-term effectiveness of tramadol in treatment of the pain of diabetic neuropathy'. Journal of Diabetes and its Complications. 14 (2): 65–70. doi:10.1016/S1056-8727(00)00060-X. PMID10959067.
- ^Barber J (2011). 'Examining the use of tramadol hydrochloride as an antidepressant'. Experimental and Clinical Psychopharmacology. 19 (2): 123–30. doi:10.1037/a0022721. PMID21463069.
- ^Göbel H, Stadler T (1997). '[Treatment of post-herpes zoster pain with tramadol. Results of an open pilot study versus clomipramine with or without levomepromazine]'. Drugs (in French). 53 Suppl 2: 34–39. doi:10.2165/00003495-199700532-00008. PMID9190323.
- ^Boureau F, Legallicier P, Kabir-Ahmadi M (July 2003). 'Tramadol in post-herpetic neuralgia: a randomized, double-blind, placebo-controlled trial'. Pain. 104 (1–2): 323–31. doi:10.1016/S0304-3959(03)00020-4. PMID12855342.
- ^Wu T, Yue X, Duan X, Luo D, Cheng Y, Tian Y, Wang K (2012). 'Efficacy and safety of tramadol for premature ejaculation: a systematic review and meta-analysis'. Urology. 80 (3): 618–24. doi:10.1016/j.urology.2012.05.035. PMID22840860.
- ^Wong BL, Malde S (2013). 'The use of tramadol 'on-demand' for premature ejaculation: a systematic review'. Urology. 81 (1): 98–103. doi:10.1016/j.urology.2012.08.037. PMID23102445.
- ^Ryan, T (2019). 'Tramadol as an adjunct to intra‐articular local anaesthetic infiltration in knee arthroscopy: a systematic review and meta‐analysis'. ANZ Journal of Surgery. doi:10.1111/ans.14920. PMID30684306.
- ^Boumendjel A, Sotoing Taïwe G, Ngo Bum E, Chabrol T, Beney C, Sinniger V, Haudecoeur R, Marcourt L, Challal S, Ferreira Queiroz E, Souard F, Le Borgne M, Lomberget T, Depaulis A, Lavaud C, Robins R, Wolfender JL, Bonaz B, De Waard M (November 2013). 'Occurrence of the Synthetic Analgesic Tramadol in an African Medicinal Plant'. Angewandte Chemie International Edition. 52 (45): 11780–84. doi:10.1002/anie.201305697. PMID24014188.
- ^ abKusari S, Tatsimo SJ, Zühlke S, Talontsi FM, Kouam SF, Spiteller M (November 2014). 'Tramadol--a true natural product?'. Angew. Chem. Int. Ed. Engl. 53 (45): 12073–6. doi:10.1002/anie.201406639. PMID25219922.
- ^Who Really did it First? Nature or a Pharmacist?, in Lab Times online; by Nicola Hunt; published September 22, 2014; retrieved November 21, 2015
- ^Kusari S, Tatsimo SJ, Zühlke S, Spiteller M (January 2016). 'Synthetic Origin of Tramadol in the Environment'. Angew. Chem. Int. Ed. Engl. 55 (1): 240–43. doi:10.1002/anie.201508646. PMID26473295.
- ^ abSouza MJ, Cox SK (2011). 'Tramadol use in zoologic medicine'. Vet Clin North Am Exot Anim Pract. 14 (1): 117–30. doi:10.1016/j.cvex.2010.09.005. PMID21074707.
External links[edit]
- Medline Plus – Patient Information Medline Plus (A Service of the U.S. National Library of Medicine)
- U.S. National Center for Biotechnology Information: Medical Genetics Summaries - Tramadol Therapy and CYP2D6 Genotype
